
Concept explainers
Interpretation:
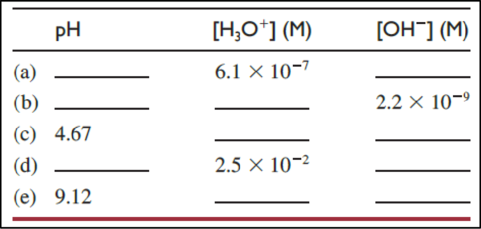
The interconversions given below have to be done and in each case whether the solution is acidic or basic has to be estimated.
Concept Introduction:
The relationship between
Other relations are given below.
If the
Explanation of Solution
(a)
The
Then,
As the
(b)
The
Then,
As the
(c)
The relationship between
Therefore,
The concentration of
As the
(d)
The
Then,
As the
(e)
The relationship between
Therefore,
The concentration of
As the
Now, the table can be filled as given below.
Now, the complete table can be given as shown below.
| Nature | |||
| (a) | Acidic | ||
| (b) | Acidic | ||
| (c) | Acidic | ||
| (d) | Acidic | ||
| (e) | Basic |
Want to see more full solutions like this?
Chapter 14 Solutions
Chemistry: The Molecular Science
- Why have chemists not tabulated the fraction ionized for different acids? Such a table would make problems such as calculating the pH of an acid solution quite simple.arrow_forwardFind the pH of solutions with the following[ H+ ]. Classify each as acidic or basic. (a) 2.7103M(b) 1.5M (c) 1.451013M (d) 6.4109Marrow_forwardFor oxyacids, how does acid strength depend on a. the strength of the bond to the acidic hydrogen atom? b. the electronegativity of the element bonded to the oxygen atom that bears the acidic hydrogen? c. the number of oxygen atoms? How does the strength of a conjugate base depend on these factors? What type of solution forms when a nonmetal oxide dissolves in water? Give an example of such an oxide. What type of solution forms when a metal oxide dissolves in water? Give an example of such an oxide.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





