
Classify each alcohol as 1°, 2°, or 3o
a.
b.
(a)
Interpretation:
The following alcohol should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to one other carbon atom is known as primary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to two other carbon atoms is known as secondary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to three other carbon atoms is known as tertiary (
Answer to Problem 23P
Primary alcohol
Explanation of Solution
The given compound is
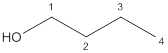
According to the above structure of compound, one hydroxyl group is present on carbon atom which is linked with one other carbon atom. Thus,
(b)
Interpretation:
The following alcohol should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to one other carbon atom is known as primary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to two other carbon atoms is known as secondary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to three other carbon atoms is known as tertiary (
Answer to Problem 23P
Tertiary alcohol
Explanation of Solution
The given compound is
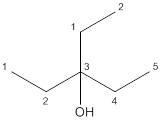
According to the above structure of compound, one hydroxyl group is present on carbon atom which is linked with three other carbon atoms. Thus,
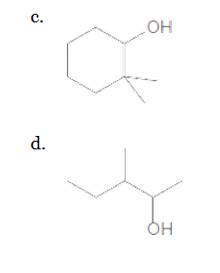
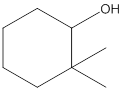
(c)
Interpretation:
The following alcohol should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to one other carbon atom is known as primary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to two other carbon atoms is known as secondary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to three other carbon atoms is known as tertiary (
Answer to Problem 23P
Secondary alcohol
Explanation of Solution
The given structure is:
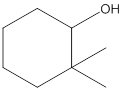
According to the above structure of compound, one hydroxyl group is present on carbon atom which is linked with two other carbon atoms. Thus, the given compound is secondary alcohol.
(d)
Interpretation:
The following alcohol should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to one other carbon atom is known as primary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to two other carbon atoms is known as secondary (
When the hydroxyl group (-OH) is present on the carbon atom and that carbon atom is bonded to three other carbon atoms is known as tertiary (
Answer to Problem 23P
Secondary alcohol
Explanation of Solution
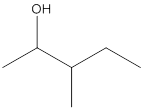
The given structure is:
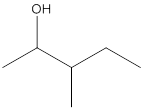
According to the above structure of compound, one hydroxyl group is present on carbon atom which is linked with two other carbon atoms. Thus, the given compound is secondary alcohol.
Want to see more full solutions like this?
Chapter 14 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- 6. Which of the following is a secondary alcohol? b. H3C -0 -CH3 OH с. О А О в O c О Е Н H3C-C-CH3 -H d. CH3OH OH H3C -C-CH3 CH3arrow_forwardR H + H3C R NaOH 95% Ethanol RT R + H2Oarrow_forwardList the products of each alcohol reaction. CH3 a. CH,-C-OH CH, NazCrO b. CH3-CH-CH;-CH2-OH c. CH-CH-OH +HCI -arrow_forward
- 2) Which of the following is a tertiary (3°) alcohol? А. D. H2 C. CH3 HO- H2C CH CH3 H2C-CH2 H3C В. OH Е. CH3 CH CH3 H3C H2 H3C- -C-CH3 С. OH H2 H3C. HO, C' H2 HCI HC HC wwwarrow_forwardList the products of each alcohol reaction. a. CH3-CH2-CH,-OH +HBr - H,S0, b. CH3-CH-CH2-OH с. СНу— CH—он +Na CH3 Na:Cr0 d. CH3-C-CH;-CH;-OH CH3arrow_forwardWhich of these compounds is a properly named 2° alcohol? a) 1,3-Propandiol HO HO b) 2-Methyl-3-methyl-3-pentanol CH3 H3C CH3 OH c) 1-Propanolarrow_forward
- 16. Identify each compound as an a cohol, a phenol, or an ether. Classify any alcohols as primary (1"), secondary (2), or tertiary (3"). a. CH,CH,CH,OH CH,CHCH, b. CHO C. CH CHOCH, CH, d.arrow_forward4. What alcohol is formed when each compound is treated with NaBH4 in MEOH? a. NaBH MeOH H3CH,CH,C H. NABH4 b. MEOH C. NaBH MEOH Section: Row: 12 Column: 4 Words: 114 O Spell Check O Local backup on Page Num: 1 Page: 1/4 1/1 MacBook Pro 411 F6 F4 esc FI F2 & %23 24 3 4. 6. R T tab Caarrow_forwardThe following compounds have similar molecular weights. Which has the highest boiling point? a.CH3CH2OH b.CH3CH=O c.CH3OCH3 d.CH3CH2CH3arrow_forward
- 7. Draw a circle around the compound in each horizontal row with the greatest water solubility and draw a rectangle around the compound with the lowest solubility in water. 1-pentanol 1-hexanol 1-octanol methanol OH Но OH Но НО 8. Classify the alcohols below as 1° or 2º or 3º. OH HO HO OHarrow_forwardQuestion 18.c of 20 Aldehydes and ketones are common carbonyl functional groups. Identify the structures and provide names of these molecules. Provide a systematic name for this structure. سکتے 5- H 1- 4- cyclo tert- 6- di n- tri 3- 2- sec- hex eth but Submit +arrow_forwardIdentify each of the following molecules an alcohol, and aldehyde or a ketone. O14 H-C-H H-C-C-CH H -C-H b. O H C-C-H 애 e,arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
