
(a)
Interpretation:
The reaction of methyl o-bromobenzoate with aqueous acid and heat has to be given.
Concept Introduction:
Hydrolysis of Ester:
The reaction in which a bond is broken by the addition of water molecule is called as hydrolysis.
Acid hydrolysis of ester:
Hydrolysis of esters yields the
The general reaction of ester hydrolysis is shown below,
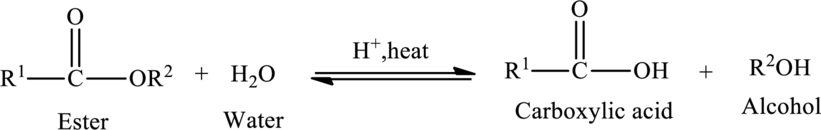
(b)
Interpretation:
The reaction of methyl o-bromobenzoate with aqueous base and heat has to be given.
Concept Introduction:
Hydrolysis of Ester:
The reaction in which a bond is broken by the addition of water molecule is called as hydrolysis.
Base hydrolysis of ester:
Hydrolysis of an ester could also be catalyzed by the presence of a strong base. The base-catalyzed hydrolysis of an ester is called as saponification.
The general reaction of base-catalyzed hydrolysis is shown below,
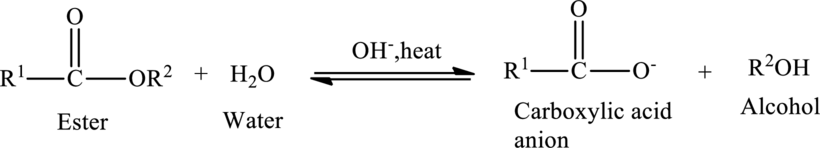
In case of base-catalyzed hydrolysis of an ester, the carboxylic acid does not exist. The reaction results in the formation of salt of carboxylic acid containing the cation of the base catalyst.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
GEN ORG/BIO CHEM LL W/CONNECT ACCESS
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





