
Concept explainers
(a)
Interpretation:
The product on reaction of
Concept introduction:
The
Answer to Problem 14.26AP
The product on reaction of
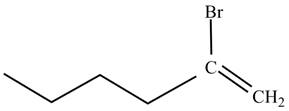
Explanation of Solution
The reaction of
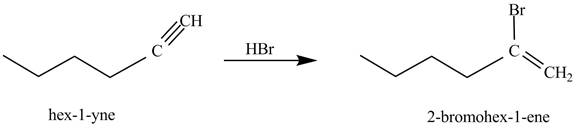
Figure 1
In the above reaction, hexyne reacts with ![]() to form a haloalkene. Therefore, the product on reaction of
to form a haloalkene. Therefore, the product on reaction of
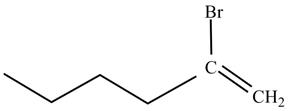
Figure 2
The product on reaction of
(b)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
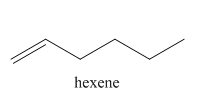
Explanation of Solution
The reaction of
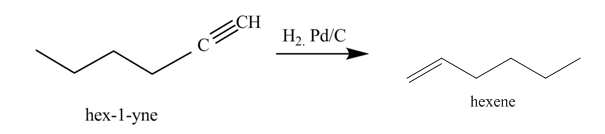
Figure 3
In the above reaction, hexyne reacts with hydrogen to form hexene. Hexyne is an unsaturated molecule consisting of a triple bond. It reacts hydrogen in presence of

Figure 4
The product on reaction of
(c)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
Explanation of Solution
The reaction of
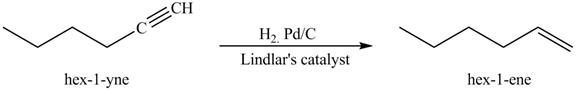
Figure 5
In the above reaction, hexyne reacts with
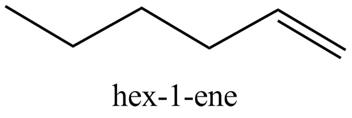
Figure 6
The product on reaction of
(d)
Interpretation:
The product on reaction of the product formed in part (c) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (c) with
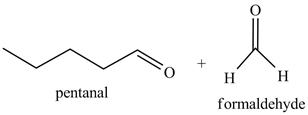
Explanation of Solution
The product formed in part (c) is shown below.
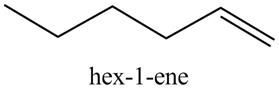
Figure 7
The reaction of the product formed in part (c) with
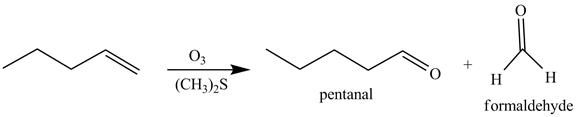
Figure 8
The above reaction is known as ozonlysis reaction. In the above reaction, a double bond is cleaved and oxidized to give two products. Reaction of hexene with

Figure 9
The products on reaction of reaction of the product formed in part (c) with
(e)
Interpretation:
The product on reaction of the product formed in part (c) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (c) with
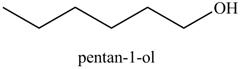
Explanation of Solution
The product formed in part (c) is shown below.

Figure 7
The reaction of above compound with
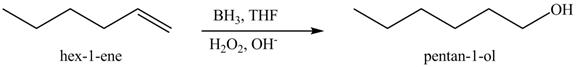
Figure 10
In the above reaction, hexene reacts with boron hydride to form organoborane which further reacts with peroxide to form an alcohol. The alcohol thus formed is by anti-markovnikov addition.

Figure 11
The product on reaction of the product formed in part (c) with
(f)
Interpretation:
The product on reaction of the product formed in part (c) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (c) with
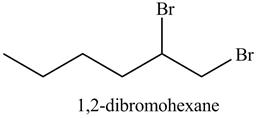
Explanation of Solution
The product formed in part (c) is shown below.
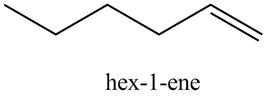
Figure 7
The reaction of above compound with
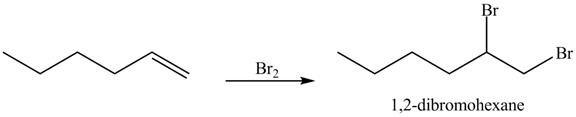
Figure 12
In the above reaction, hexene being unsaturated reacts with bromine molecule to form a dibromoalkane, that is
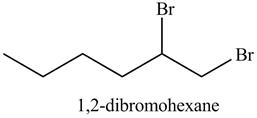
Figure 13
The product on reaction of the product formed in part (c) with
(g)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
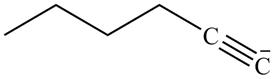
Explanation of Solution
The reaction of
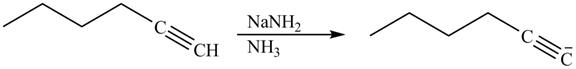
Figure 14
Alkynes react with sodamide

Figure 15
The product on reaction of
(h)
Interpretation:
The product on reaction of the product formed in part (g) with
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (g) with
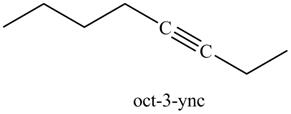
Explanation of Solution
The product formed in part (g) is shown below.
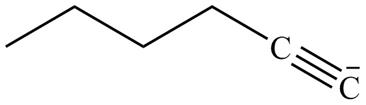
Figure 15
The reaction of above compound with
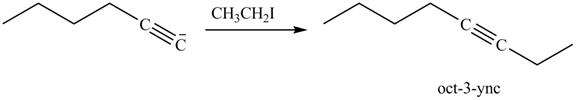
Figure 16
In the above reaction, the anion of alkyne obtained reacts with iodoethane to form an alkyne of eight carbons. It is non-terminal alkyne, that is, triple bond is not situated at the terminal end. The product formed is shown below.
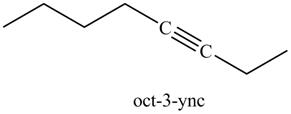
Figure 17
The product on reaction of the product formed in part (g) with
(i)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
Explanation of Solution
The reaction of
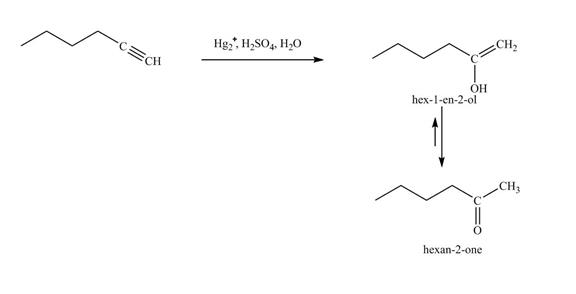
Figure 18
In the above reaction, hexyne reacts with

Figure 19
The product on reaction of
(j)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of
Explanation of Solution
The reaction of
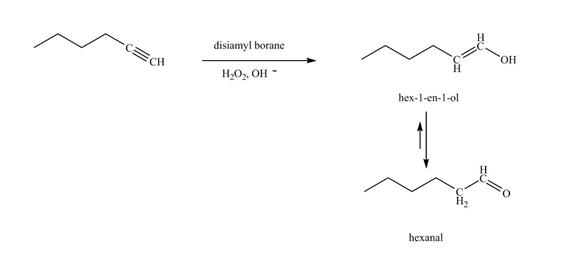
Figure 20
In the above reaction, hexyne reacts with disiamyl borane to form an organoborane. This reaction is also known as hydroboration-oxidation reaction. The organo-borane gives an enol which tautomerism to form an
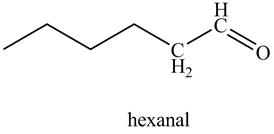
Figure 21
The product on reaction of
(k)
Interpretation:
The product on reaction of
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The products formed on reaction of
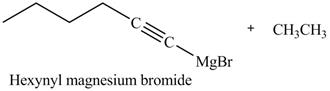
Explanation of Solution
The reaction of
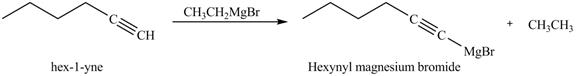
Figure 22
In the above reaction,

Figure 23
The products formed on reaction of
(l)
Interpretation:
The product on reaction of the product formed in part (k) with ethylene oxide and
Concept introduction:
The alkynes consist of a triple bond between two carbon atoms. The general formula of alkynes is
Answer to Problem 14.26AP
The product on reaction of the product formed in part (k) with ethylene oxide and
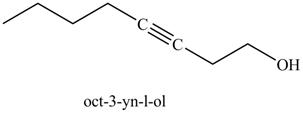
Explanation of Solution
The product formed in part (k) is shown below.
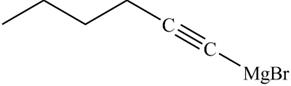
Figure 23
The reaction of above compound with ethylene oxide and
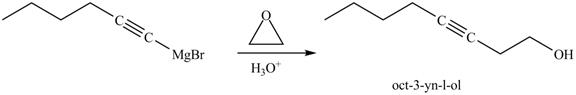
Figure 24
In the above reaction, the Grignard reagent obtained in part (k) reacts with ethylene oxide followed by hydrolysis to form an alcohol with a triple bond in between the chain. The product formed on reaction of product formed in part (k) with ethylene oxide and
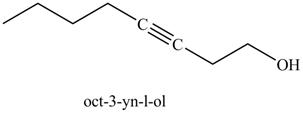
Figure 25
The product on reaction of the product formed in part (k) with ethylene oxide and
Want to see more full solutions like this?
Chapter 14 Solutions
EBK ORGANIC CHEMISTRY
- Compound X (C4H9Br) reacts by heating with NaOH in H2O to form Y. The compound Y then undergoes acid catalysed hydration by H2SO4 in 180°C to form 2-methyl prop-1-ene. (e) Determine the structure of X and Y. (f) Predict a MAJOR product when compound Y reacts with H2SO4 in 140°C. (g) Draw a structural isomer of X. Name the isomer using IUPAC nomenclature. (h) Describe a chemical test to distinguish between compound Y and 1-butanol.arrow_forwardGive reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.arrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward
- Draw a structural formula for the product formed by treating each compound with warm chromic acid, H2CrO4:(a) CH3(CH2)4CH2OH (b) (Picture attached) Please name both of the products, thanks!arrow_forward(a) A hydrocarbon isolated from fish oil and from plankton was identified as 2,6,10,14-tetramethyl-2-pentadecene. Write its structure.(b) Alkyl isothiocyanates are compounds of the type RN C S. Write a structural formula for allyl isothiocyanate, a pungent-smelling compound isolated from mustard.(c) Grandisol is one component of the sex attractant of the boll weevil. Write a structural formula for grandisol given that R in the structure shown is an isopropenyl group.arrow_forwardGive reasons for the following :(i) Phenol is more acidic than methanol.(ii) The C—O—H bond angle in alcohols is slightly less than the tetrahedral angle (190°28′).(iii) (CH3)3C—O—CH3 on reaction with HI gives (CH3)3C—I and CH3—OH as the main products and not (CH3)3C—OH and CH3—I.arrow_forward
- Predict the major products (including stereochemistry) when cis-3-methylcyclohexanol reacts with the following reagents. (a) concentrated HBr (b) TsCl/pyridine, then NaBrarrow_forward(a) Why is the following reaction a poor method for the synthesis of t-butyl propyl ether? (b) What would be the major product from this reaction? (c) Propose a better synthesis of r-butyl propyl ether. ÇH, does not give ÇH, CH,CH,CH,-O: *Na + CH,-C-Br > CH,-C-o-CH,CH,CH, CH, CH, t-butyl propyl ether sodium propoxide 1-butyl bromidearrow_forward1. Deduce the structure of each compound from the information given. All unknowns in this problem have molecular formula C8H12. (a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC—(CH2)6—COOH. Draw the structure of W. (b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethyl sulfide, gives two equivalents of butanedial, O=CH—CH2CH2—CH=O. Draw the structure of X.arrow_forward
- Draw the reaction products of the following reactions of the compound 1-pentanol and name the compounds according to IUPAC! (a) B-elimination of H20 (b) SN reaction in the presence of hydrochloric acid (c) Condensation with acetic acid (d) Oxidation to the corresponding carbonyl compound (e) Condensation of two butanol moleculesarrow_forwardThe reaction of 1-bromopropane and sodium hydroxide in ethanol occurs by an SN2mechanism. What happens to the rate of this reaction under the following conditions?(a) The concentration of NaOH is doubled.(b) The concentrations of both NaOH and 1-bromopropane are doubled.(c) The volume of the solution in which the reaction is carried out is doubled.arrow_forward(1) NaOH, H₂O (2) LiAlH (3) H₂O (S)-2-methyl-pentane-2,4-diol What type of isomerism does the product have?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





