
Concept explainers
How many absorptions would you expect to observe in the 13C NMR spectra of the following compounds?
(a) 1,1-Dimethylcyclohexane
(b) CH3CH2OCH3
(c) tert-Butylcyclohexane
(d) 3-Methyl-l-pentyne
(e) cis-1,2-Dimethylcyclohexane
(f) Cyclohexanone
a)
Interpretation:
The structure of an given molecular formula C8H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
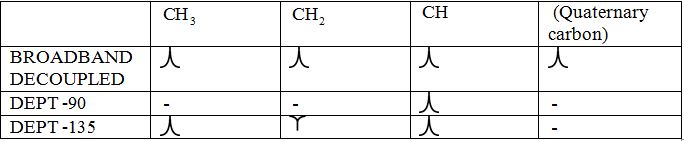
Broadband-decoupled 13CNMR spectrum: The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
i) DEPT -90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
ii) DEPT -135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:
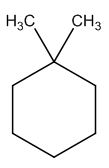
To identify:
The structure of given molecular formula C8H16.
Answer to Problem 47AP
Explanation of Solution
Calculate HDI value:
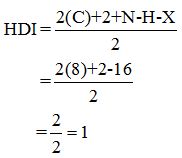
The HDI calculation led to confirm about the presence of a double bond or a ring.
Interpret the given 13CNMR spectrum:
Broadband - decoupled spectrum:
The spectrum shows five signals whereas the given molecular formula also has eight carbon atoms. Thus all the eight carbons have chemically different electronic environments showing signals.
i) The five signals in the region of 0-50ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene or Methine groups.
DEPT -90: Spectrum has no signals led to confirmation of no CH group in the structure.
DEPT -135 (gives signals of CH2, CH3 and C groups):
ii) The two positive signals indicate the presence of two methyl groups as no signals appear in the DEPT -90 spectrum.
iii) The three negative signals indicate the presence of four methylene groups; only methylene groups appear negative in the spectrum.
iv) So far the group of fragments obtained is
Two -CH3, -C-, four -CH2
From the fragments (CH3 + CH3 + (CH2)5 + C = 16 protons) sixteen protons are obtained whereas the total number of protons from the molecular formula is also sixteen.
The possible structures are:
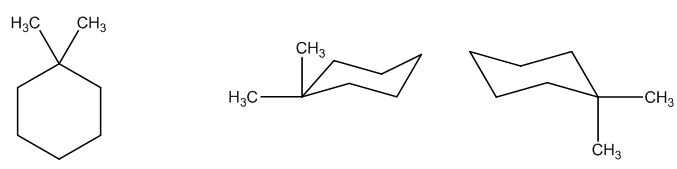
The first structure possesses rotational symmetry and exhibits fewer signals in the spectrum and gets cancelled out.

The structure of a given molecular formula C8H16 is predicted using 13CNMR spectra.
b)
Interpretation:
The structure of an given molecular formula C8H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
Broadband-decoupled 13CNMR spectrum: The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
i) DEPT -90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
ii) DEPT -135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:

To identify:
The structure of given molecular formula C3H8O.
Answer to Problem 47AP
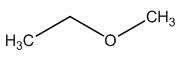
Explanation of Solution
Calculate HDI:
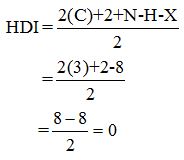
The HDI calculation led to confirm about the absence of a double bond or a ring.
Interpret the given 13CNMR spectrum:
Broadband - decoupled spectrum:
The spectrum shows three signals whereas the given molecular formula also has three carbon atoms. Thus all the three carbons have chemically different electronic environments showing signals.
i) The five signals in the region of 0-50ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene groups.
DEPT -90: Spectrum has no signals led to confirmation of no CH group in the structure.
DEPT -135 (gives signals of CH2, CH3 groups):
ii) The two positive signals indicate the presence of two methyl groups as no signals appear in the DEPT -90 spectrum.
iii) The spectrum shows three signals whereas the given molecular formula also has three carbon atoms. Thus all the three carbons have chemically different electronic environments showing signals.
iv) So far the group of fragments obtained is
Two -CH3, -CH2
From the fragments (CH3 + CH3 + CH2 = 8 protons) eight protons are obtained whereas the total number of protons from the molecular formula is also eight.
The structure of a given molecular formula C3H8O is predicted using 13CNMR spectra.
c)
Interpretation:
The structure of an given molecular formula C8H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
Broadband-decoupled 13CNMR spectrum: The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
i) DEPT -90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
ii) DEPT -135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:

To identify:
The structure of given molecular formula C10H20.
Answer to Problem 47AP
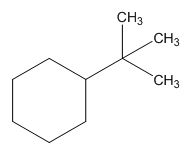
Explanation of Solution
Calculate HDI:
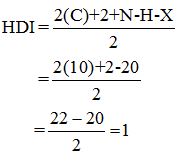
The HDI calculation led to confirm about the presence of a double bond or a ring.
Interpret the given 13CNMR spectrum:
Broadband - decoupled spectrum:
The spectrum shows six signals whereas the given molecular formula also has ten carbon atoms. Thus all the ten carbons have chemically different electronic environments showing signals.
i) The two signals in the region of 0-50ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene groups.
DEPT -90: Spectrum has signals led to confirmation of CH group in the structure.
DEPT -135 (gives signals of CH2, C, CH3 groups):
ii) The two positive signals indicate the presence of three methyl groups as no signals appear in the DEPT -90 spectrum.
iii) The three negative signals indicate the presence of five methylene groups; only methylene groups appear negative in the spectrum.
iv) So far the group of fragments obtained is
Two -CH3, -CH2
From the fragments (CH3)3 + (CH2)5 + CH + C = 20 protons) twenty protons are obtained whereas the total number of protons from the molecular formula is also twenty.
The structure of a given molecular formula C10H20 is predicted using 13CNMR spectra.
d)
Interpretation:
The structure of an given molecular formula C8H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
Broadband-decoupled 13CNMR spectrum: The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
i) DEPT -90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
ii) DEPT -135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:

To identify:
The structure of given molecular formula C6H10.
Answer to Problem 47AP
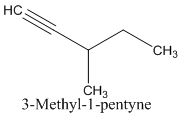
Explanation of Solution
Calculate HDI:
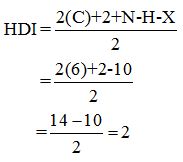
The HDI calculation led to confirm about the presence of a double bond or a ring. (two unsaturated degree).
Interpret the given 13CNMR spectrum:
Broadband - decoupled spectrum:
The spectrum shows six signals whereas the given molecular formula also has six carbon atoms. Thus all the six carbons have chemically different electronic environments showing signals.
i) The two signals in the region of 0-50ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene groups.
DEPT -90: Spectrum has signals led to confirmation of CH group in the structure.
DEPT -135 (gives signals of CH2, C, CH3 groups):
ii) The four positive signals indicate the presence of two methyl groups as no signals appear in the DEPT -90 spectrum.
iii) The negative signals indicate the presence of methylene groups; only methylene groups appear negative in the spectrum.
iv) So far the group of fragments obtained is
Two -CH3, -CH2
From the fragments (CH3)2 + CH2 + (CH)2 + C = 10 protons) ten protons are obtained whereas the total number of protons from the molecular formula is also ten.
The structure of a given molecular formula C6H10 is predicted using 13CNMR spectra.
e)
Interpretation:
The structure of an given molecular formula C8H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
Broadband-decoupled 13CNMR spectrum: The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
i) DEPT -90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
ii) DEPT -135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:

To identify:
The structure of given molecular formula C8H16.
Answer to Problem 47AP
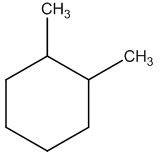
Explanation of Solution
Calculate HDI:
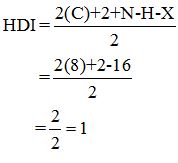
The HDI calculation led to confirm about the presence of a double bond or a ring.
Interpret the given 13CNMR spectrum:
Broadband - decoupled spectrum:
The spectrum shows four signals whereas the given molecular formula also has eight carbon atoms. Thus all the eight carbons have chemically different electronic environments showing signals.
i) The four signals in the region of 0-50ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene or Methine groups.
DEPT -90: Spectrum has no signals led to confirmation of CH group in the structure.
DEPT -135 (gives signals of CH2, CH3 and CH groups):
ii) The two positive signals indicate the presence of two methyl groups as no signals appear in the DEPT -90 spectrum.
iii) The two negative signals indicate the presence of four methylene groups; only methylene groups appear negative in the spectrum.
iv) So far the group of fragments obtained is
Two -CH3, -CH-, four -CH2
From the fragments (CH3 + CH3 + (CH2)4 + (CH)2 = 16 protons) sixteen protons are obtained whereas the total number of protons from the molecular formula is also sixteen.
The structure of a given molecular formula C8H16 is predicted using 13CNMR spectra.
f)
Interpretation:
The structure of an given molecular formula C8H16 to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C - 13C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
Broadband-decoupled 13CNMR spectrum: The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
i) DEPT -90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
ii) DEPT -135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:

To identify:
The structure of given molecular formula C6H10O.
Answer to Problem 47AP
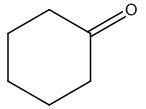
Explanation of Solution
Calculate HDI:
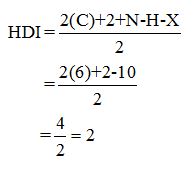
The HDI calculation led to confirm about the presence of a double bond or a ring.
Interpret the given 13CNMR spectrum:
Broadband - decoupled spectrum:
The spectrum shows four signals whereas the given molecular formula also has six carbon atoms. Thus all the six carbons have chemically different electronic environments showing signals.
i) The four signals in the region of 0-50ppm indicate the sp3 hybridized carbon atoms which can be methyl / methylene or Methine groups.
DEPT -135 (gives signals of CH2, C groups):
ii) The two negative signals indicate the presence of four methylene groups; only methylene groups appear negative in the spectrum.
iii) So far the group of fragments obtained is
C=O, -C-, four -ch2
From the fragments (CH2)5 + C = 10 protons) ten protons are obtained whereas the total number of protons from the molecular formula is also ten.
The structure of a given molecular formula C6H10O is predicted using 13CNMR spectra.
Want to see more full solutions like this?
Chapter 13 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Ketones undergo a reduction when treated with sodium borohydride, NaBH4. What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm-1 and M+=74 in the mass spectrum?arrow_forwardHow would you use NMR (either 13C or 1H) to distinguish between the following pairs of isomers?arrow_forwardCompound H (C8H6O3) gives a precipitate when treated with hydroxylamine in aqueous ethanol and a silver mirror when treated with Tollens solution. Following is its 1H-NMR spectrum. Deduce the structure of compound H.arrow_forward
- Propose a structural formula for the analgesic phenacetin, molecular formula C10H13NO2, based on its 1H-NMR spectrum.arrow_forwardBased on the following 1H-NMR data, what is the possible structure of the alcohol C5H11OH?arrow_forwardIn the 1H NMR spectra of 2-bromopropane (CH3)2CHBr and 1-bromopropane CH3CH2CH2Br, how many signals do you expect to see?arrow_forward
- A hydrocarbon, compound B, has molecular formula C6H6, and gave an NMR spectrum with two signals: delta 6.55 pm and delta 3.84 pm with peak ratio of 2:1. When warmed in pyridine for three hr, compound B quantitatively converts to benzene. Mild hydrogenation of B yielded another compound C with mass spectrum of m/z 82. Infrared spectrum showed no double bonds; NMR spectrum showed one broad peak at delta 2.34 ppm. With this information, address the following questions. a) How many rings are in compound C? b) How many rings are probably in B? How many double bonds are in B? c) Can you suggest a structure for compounds B and C? d) In the NMR spectrum of B, the up-field signal was a quintet, and the down field signal was a triplet. How must you account for these splitting patterns?arrow_forwardPredict the theoretical number of different NMR signals produced by each compound, and give approximate chemical shifts. Point out any diastereotopic relationships. (a) 2-bromobutane (b) cyclopentanolarrow_forwardThe H1H1 NMR spectra corresponds to an alcohol with the molecular formula C5H12O. Deduce the structure from the spectraarrow_forward
- Draw the structure of molecular formula C8H10O that produced the 1H NMR spectra shown below. The IR spectrum does not show a broad absorbance at 3300 cm–1 or a strong absorbance at 1710 cm–1.arrow_forwardThe following ¹H NMR spectrum is shown in two questions. "nmrsim presentation" 1 1 C:\Bruker TopSpin3.5p17\examdata 10 1,4-Disubstituted benzene ring Which functional group is BEST attributed to the pair of doublets at about 7-8 ppm, each integrating to 2H? trans-alkene Cyclohexene Carboxylic acid [ppm] 1,3-Disubstituted benzene ring [oa]arrow_forwardThe mass spectrum of a compound with a molecular formula C9H10O is shown below. Which compound would you expect to give rise to the mass spectra shown below? A) Benzyl methyl ketone B) 3-Methyl-2,3-dihydro-benzofuran C) 2-Allylphenol D) Benzenepropanal E) 1-bromo-1-butenearrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

