
(a)
Interpretation:
The melting and boiling point temperature of acetic acid in below graph has to be identified.
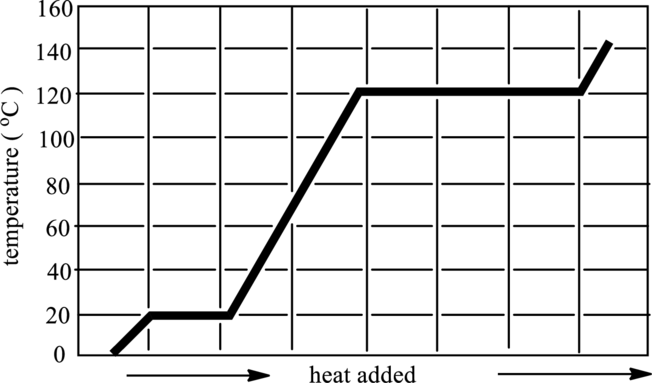
Concept Introduction:
A particle in the substance that occupies space and has mass is termed as matter. Atoms and molecules are composed of matter. There are three types of
1 Solid: In solid, particles are tightly packed and are highly dense.
2 Liquid: In liquid, particles are loosely packed and are less dense.
3 Gases: In gases, particles move freely with no force of attraction.
(b)
Interpretation:
The graph that indicates solid, liquid and gaseous phase in below graph has to be redrawn.
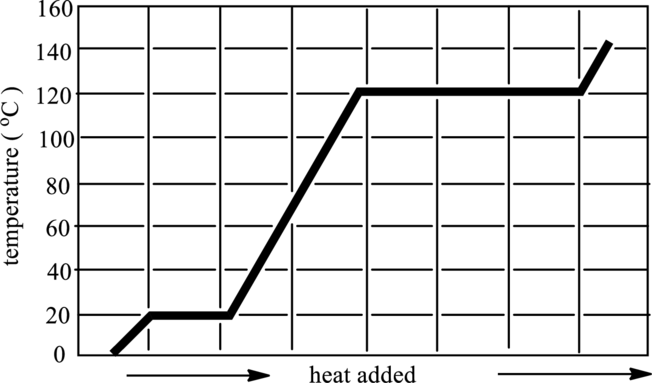
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
Regions of transition in states have to be indicated in below graph. Also, phases present in it has to be determined.
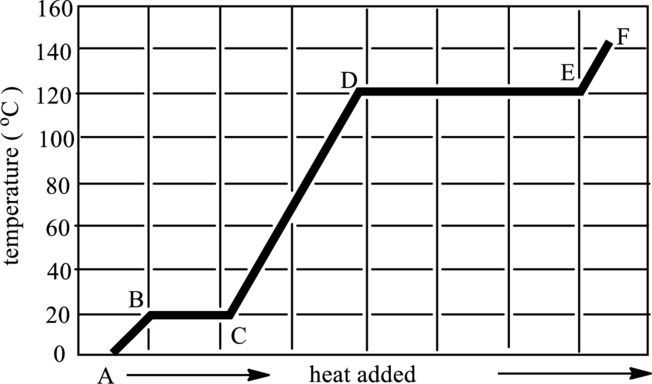
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Foundations of College Chemistry 15e Binder Ready Version + WileyPLUS Registration Card
- Define the joule in terms of SI base units.arrow_forwardIf 14.5 kJ of heat were added to 485 g of liquid water, how much would its temperature increase?arrow_forwardA burning match and a bonfire may have the same temperature, yet you would not sit around a burning match on a fall evening to stay warm. Why not?arrow_forward
- 2. Why does the temperature of a mixture of ice and water remain constant during melting and freezing?arrow_forwardCalcium chloride is an ice melting compound used for sidewalks and streets. This is an exothermic reaction in which calcium chloride reacts with water which releases heat to the surroundings causing the ice to melt. Using the language of breaking and making bonds and the energy required/released when that happens, how can you describe the temperature change observed when calcium chloride is dissolved in water?arrow_forward5. Describe the phase of ice and water vapor in terms of the function of temperature and pressure.arrow_forward
- 3. Is solid > liquid → gas an endothermic or exothermic process? Describe how this energy change affects the energy of the molecules and their intermolecular forces.arrow_forward3. True or false: Because dry ice sublimes, carbon dioxide has no liquid phase. (a) True (b) Falsearrow_forwardHow much energy is given off when 563.8 g of NaCl solidifies at its freezing point of 801C ?arrow_forward
- 2. As energy is added to a substance, the temperature remains constant. How may the substance be changing? ,arrow_forwardAs water cools to a temperature of zero degrees Celsius and forms ice, water molecules tend to move farther apart. vībrate rapidly. flow more randomly. gradually expand.arrow_forwardThere are three sets of sketches below, showing the same pure molecular compound (ammonia, molecular formula NH₂) at three different temperatures. The sketches are drawn as if a sample of ammonia were under a microscope so powerful that individual atoms could be seen. Only one sketch in each set is correct. Use the slider to choose the correct sketch in each set. You may need the following information: melting point of NH3: -77.7 °C boiling point of NH₁: -33.3 °C B (Choose one) (Choose one) (Choose one) -59, °C -89, °C 54, °Carrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





