
EBK ESSENTIAL ORGANIC CHEMISTRY
3rd Edition
ISBN: 9780100659469
Author: Bruice
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13, Problem 29P
(a)
Interpretation Introduction
Interpretation:
Aldehyde or ketone that is obtained when the given compound is heated in basic aqueous solution to be identified.
Concept introduction:
Aldol reaction is an addition reaction of
Mechanism for the aldol addition:
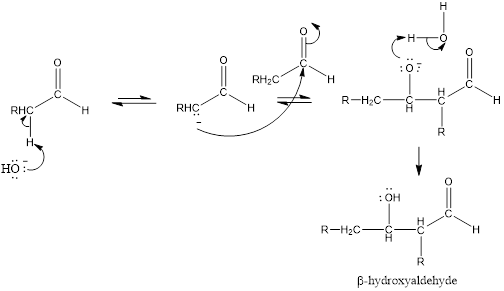
(b)
Interpretation Introduction
Interpretation:
Aldehyde or ketone that is obtained when the given compound is heated in basic aqueous solution to be identified.
Concept introduction:
Aldol reaction is an addition reaction of aldehydes and ketones.
Mechanism for the aldol addition:
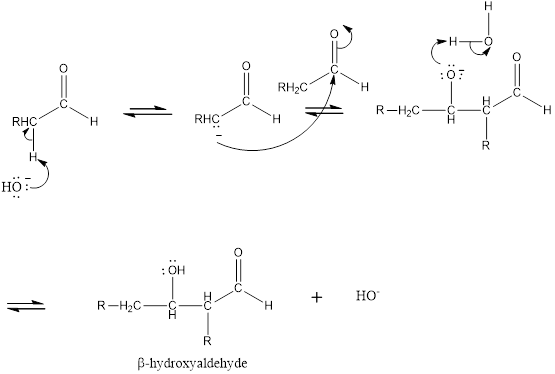
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following is also an acceptable name for 3-nitrophenol?
3-cresol
p-nitrophenol
o-nitrophenol
m-nitrophenol
hydroquinone
The result of the condensation reaction of aldehydes with phenols is:
A) Arylmethane (auric) dye
B) Indophenol dye
C) azo dye
D) Substituted benzophenone
Draw the pyrrole that would form in each of the following reactions.
a)
b)
COOEt
NH3
Ph
cat HCI, heat
NH2
cat HCI, heat
c)
2 equiv
NH2
Chapter 13 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
Ch. 13.1 - Identify the most acidic hydrogen in each...Ch. 13.1 - Prob. 2PCh. 13.1 - Prob. 3PCh. 13.1 - Prob. 4PCh. 13.1 - Explain why HO cannot remove a proton from the...Ch. 13.2 - Prob. 6PCh. 13.2 - Prob. 7PCh. 13.3 - Prob. 8PCh. 13.3 - Prob. 9PCh. 13.3 - Prob. 10P
Ch. 13.4 - Prob. 11PCh. 13.5 - Prob. 12PCh. 13.5 - Prob. 13PCh. 13.6 - Prob. 14PCh. 13.7 - Prob. 16PCh. 13.8 - Prob. 17PCh. 13.8 - Prob. 18PCh. 13.8 - Prob. 19PCh. 13.9 - Prob. 20PCh. 13.10 - Propose a mechanism for the formation of...Ch. 13.10 - Prob. 22PCh. 13.10 - a. If the biosynthesis of palmitic acid were...Ch. 13 - Draw the enol tautomers for each of the following...Ch. 13 - Number the following compounds in order from...Ch. 13 - Prob. 26PCh. 13 - Explain why the pKa of a hydrogen bonded to the...Ch. 13 - Prob. 28PCh. 13 - Prob. 29PCh. 13 - Prob. 30PCh. 13 - Prob. 31PCh. 13 - Prob. 32PCh. 13 - Prob. 33PCh. 13 - Using cyclopentanone as the reactant, show the...Ch. 13 - Prob. 35PCh. 13 - Prob. 36PCh. 13 - Prob. 37PCh. 13 - Prob. 38PCh. 13 - Prob. 39PCh. 13 - Prob. 40PCh. 13 - Prob. 41PCh. 13 - Prob. 42PCh. 13 - Prob. 43PCh. 13 - Prob. 44PCh. 13 - Describe how the following compounds can be...Ch. 13 - Prob. 46PCh. 13 - Which would require a higher temperature:...Ch. 13 - Prob. 48PCh. 13 - Propose a mechanism for the following reaction:Ch. 13 - Show how the following compounds could be...Ch. 13 - Prob. 51PCh. 13 - Prob. 52P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the IUPAC name for the following compound? O t-butyl cyclohexanecarboxylate O 3-cyclohexyl-2-oxy-tert-butanone O butyl cyclohexanone O t-butyl cyclohexanone O butyl cyclohexanecarboxylatearrow_forwardWhat is the reduction product of the following compound with H2/Pd? A 2-propyl-1-cyclohexanol B 2-propenyl-1-cyclohexanol C2-propylcyclohexanone D 1-propyl-2-cyclohexanol What is the oxidation product of this compound? H' A ethanoic acid acetic acid propanoic acid D propanone What is the oxidation product of this compound? A 3,4-dimethylpentanoic acid B 2,3-dimethylpentanoic acid C2,3-dimethylpentanone 3,4-dimethylpentanonearrow_forwardWhat two functional groups react to form the following? a. A hemiacetal b. An acetal c. A ketal d. A hemiketalarrow_forward
- The following molecule belongs to a class of compounds called enediols; each carbon of the double bond carries an OH group. Draw structural formulas for the -hydroxyketone and the -hydroxyaldehyde with which this enediol is in equilibrium.arrow_forwardLabel each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forwardWhat is the IUPAC name for the following compound? CI 4-ethyl-3,5-dimethylhexanoyl chloride O 3-ethyl-2,4-dimethylhexanoyl chloride 4-ethyl-3,5-dimethylpentanoyl chloride O 3-ethyl-2,4-dimethylpentanoyl chloridearrow_forward
- What is the IUPAC name of the following compound? 4-phenyl-3-hexanone O 3-phenyl-4-hexanone O 1,2-diethyl-2-phenylethanal O 1-methyl-1-phenyl-2-butanonearrow_forwardGive the products formed when Benzaldehyde and Benzoic Acid are treated with the given reagents. - NH2OHarrow_forwardWhat is the common name for the aldehyde with molecular formula CH2O? A) hydroaldehyde B) benzaldehyde C) formaldehyde D) methane oxide A Вarrow_forward
- What is the final product when you mix 4-nitrobenzaldehyde with each of these: a) 4-bromophenylthydrazine hydrochloride b) 4-cyanophenylhydrazine hydrochloride c) aminoguanidine bicarbonate d) 4-chlorophenylhydrazine hydrochloridearrow_forwardPredict the products formed when cyclohexanone reacts with the following reagents. ) Tollens reagentarrow_forwardThe compound shown is the principal component of vanilla bean extracts: H HO What is the IUPAC name of this compound? O 1-formyl-3-methoxyphenol O 3-formyl-2-methoxyphenol O 4-hydroxy-3-methoxybenzaldehyde O 1-hydroxy-2-methoxybenzaldehyde O None of these OCH 3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY