
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.58AQAP
Draw the condensed or line-angle structural formula for the
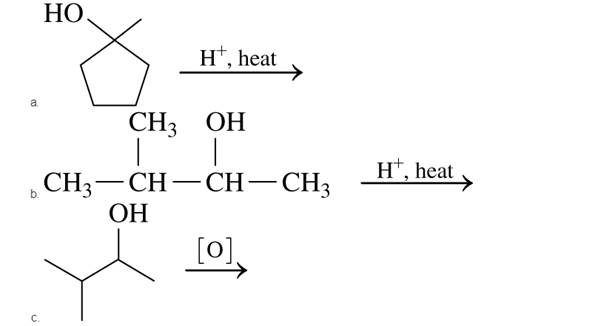
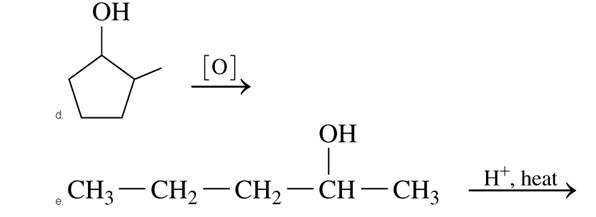
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10. Draw the structural formula for the product in each of the following addition reactions for an alkene: (11.7)
a) CH3-CH2-C=CH-CH3 + H,O
CH3-CH₂
b)
CH 3
CH3-CH-C=CH-CH3 + H₂
CH3
+ H₂
Pt
H
Ni
12.60 Which of the following aldehydes or ketones are soluble in
water? (12.3)
|3|
a. CH3-CH2 -C-CH3
b. CH3 — С —н
CH3
c. CH3-CH2-CH-CH2-CH2-C-H
12.50 Draw the condensed or line-angle structural formula, if cyclic,
for each of the following compounds: (12.1, 12.2)
a. 2,3-dichlorocyclobutyl alcohol
C. 2-methyl-5-bromo-3-hexanol
b. butyl propyl ether
d. 2-methyl-4-nitrophenol
Chapter 13 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 13.1 - Prob. 13.1QAPCh. 13.1 - Prob. 13.2QAPCh. 13.1 - Prob. 13.3QAPCh. 13.1 - Prob. 13.4QAPCh. 13.2 - Prob. 13.5QAPCh. 13.2 - Prob. 13.6QAPCh. 13.2 - Prob. 13.7QAPCh. 13.2 - Prob. 13.8QAPCh. 13.3 - Prob. 13.9QAPCh. 13.3 - Prob. 13.10QAP
Ch. 13.3 - Prob. 13.11QAPCh. 13.3 - Prob. 13.12QAPCh. 13.3 - Prob. 13.13QAPCh. 13.3 - Prob. 13.14QAPCh. 13.3 - Prob. 13.15QAPCh. 13.3 - Prob. 13.16QAPCh. 13.4 - Prob. 13.17QAPCh. 13.4 - Prob. 13.18QAPCh. 13.4 - Prob. 13.19QAPCh. 13.4 - Prob. 13.20QAPCh. 13.4 - Prob. 13.21QAPCh. 13.4 - Prob. 13.22QAPCh. 13.4 - Prob. 13.23QAPCh. 13.4 - Prob. 13.24QAPCh. 13.4 - Prob. 13.25QAPCh. 13.4 - Prob. 13.26QAPCh. 13.4 - a. Which of the functional groups alkene alcohol,...Ch. 13.4 - Prob. 13.28QAPCh. 13 - Prob. 13.29UTCCh. 13 - 13.30 Identify each of the following as an...Ch. 13 - Prob. 13.31UTCCh. 13 - Prob. 13.32UTCCh. 13 - Prob. 13.33UTCCh. 13 - Prob. 13.34UTCCh. 13 - Prob. 13.35UTCCh. 13 - Prob. 13.36UTCCh. 13 - Prob. 13.37AQAPCh. 13 - Prob. 13.38AQAPCh. 13 - Prob. 13.39AQAPCh. 13 - Prob. 13.40AQAPCh. 13 - Prob. 13.41AQAPCh. 13 - Prob. 13.42AQAPCh. 13 - Prob. 13.43AQAPCh. 13 - Prob. 13.44AQAPCh. 13 - Prob. 13.45AQAPCh. 13 - Prob. 13.46AQAPCh. 13 - Prob. 13.47AQAPCh. 13 - Prob. 13.48AQAPCh. 13 - Explain why each of the following compounds would...Ch. 13 - Prob. 13.50AQAPCh. 13 - Prob. 13.51AQAPCh. 13 - Prob. 13.52AQAPCh. 13 - Prob. 13.53AQAPCh. 13 - Prob. 13.54AQAPCh. 13 - Prob. 13.55AQAPCh. 13 - Dimethyl ether and ethyl alcohol both have the...Ch. 13 - Prob. 13.57AQAPCh. 13 - Draw the condensed or line-angle structural...Ch. 13 - Prob. 13.59AQAPCh. 13 - Menthol, which has a minty flavor, is used in...Ch. 13 - Prob. 13.61CQCh. 13 - Prob. 13.62CQCh. 13 - Prob. 13.63CQCh. 13 - Prob. 13.64CQCh. 13 - Prob. 13.65CQCh. 13 - Prob. 13.66CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (12.3)Which of the following has the strongest dispersion force between its molecules? O CH3CH3 O CH3CH₂CH₂CH₂CH3 O All of these have the dispersion forces with the same strength. O CH3CH₂CH₂CH3 O CH3CH₂CH₂CH₂CH₂CH3arrow_forward12.38 The compound frambinone has the taste of raspberries and has been used in weight loss. Identify the functional groups in frambinone. (12.1, 12.3) HO O CH,—CH,—C–CH3 14 Frambinone odioarrow_forwardS 4 Draw all possible isomers for the chemical formula C₂H₂O. Select 2 000 000 → % 5 C Draw H 6 O Rings MacBook Pro & 7 * 8 More ( 9 ) 0arrow_forward
- 11.10 Draw the condensed structural formula for alkanes or the line- angle formula for cycloalkanes for each of the following: ommo) emo2 to zslumot IS a. propane b. hexane c. heptane d. cyclohexane onstudolay the ends leboM onsqongolaarrow_forwardWhat is the IUPAC name of the following compound? O bicyclo[6.5.4]undecane bicyclo[4.3.2]nonane [4.3.2]bicyclononane O [2.3.4]bicycloundecane O bicyclo[4.3.2]undecane Aarrow_forwardConvert the structure to IUPAC name or IUPAC name to structure, as appropriate. Structure IUPAC Name 5-methylhex-4-en-2-ol 6-bromobicyclo[3.2.2]nonanearrow_forward
- Read carefully the given partial names in IUPAC alkane nomenclature and indicate whether its Accurate, Incorrect, or Impossible Pentylbutane Spiro[6.3]decane Cycloethylmethane Butylpentane Tetramethylethane Cycloethyl Cyclobutylarrow_forwardQ2. Using a series of steps, show how you would synthesize N-ethylpropanamide. You are given propanal and ethanamine to begin with. You may use any other reactants or catalysts that you need. The steps should be chemical reactions with the structures of the reactants and products drawn out. (10T)arrow_forward11.2 Identify each of the following as a formula of an organic or inorganic compound. For an organic compound, indicate if represented as molecular formula, expanded, or condensed structural formula: a. C6H12O6 c. 1₂ 2sols b. K₂PO4 H d. H-C-S-H e. CH3-CH₂-CH₂-CH₂-CH3 f. C4H,Br Harrow_forward
- 11.34 Draw the structural formula for the product in each of the fol- lowing reactions: a. CH3–CH,—CH=CH, + HO b. C. d. + H₂ + H₂ Pt ✓ + H₂O H Harrow_forward4.15 Write the skeletal structure for the alkane or cycloalkane shown: a. C6H12 b. CH3CH₂CH₂CH3 c. CH3CH₂CH₂CH₂CH₂CH₂CH₂CH3arrow_forwardSubstitution reactions of benzene (13.13)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License