
Interpretation:
The
Concept introduction:
The group of symmetry operations of which atleast one point is kept fixed is called point group. The symmetry operations can be identity, rotation, reflection, inversion and improper rotation.
Answer to Problem 13.43E
The
The coordinates for hydrogen in
Explanation of Solution
The structure of ammonia molecule is,
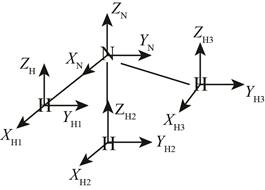
Figure 1
The total coordinates in the given system is
The ammonia molecule has non-linear structure and
On applying the
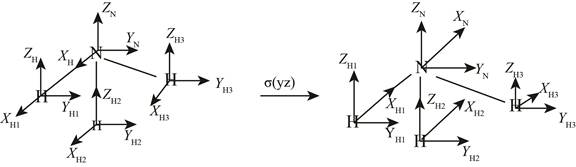
Figure 2
The
Similarly on applying the
The
Similarly on applying the
The
The coordinates for hydrogen for
The
The coordinates for hydrogen in
Want to see more full solutions like this?
Chapter 13 Solutions
Bundle: Physical Chemistry, 2nd + Student Solutions Manual
- Construct the Hckel determinants for cyclobutadiene and cyclopentadiene. In what ways are they alike? In what ways are they different?arrow_forwardIdentify the symmetry elements present in the following objects. a The Eiffel Tower. You may have to look up a picture of it if you dont remember its shape b Any book ignore the printing. c An octagonal wood block. d A jack from the set of jacks pictured here: Note that some of the points end differently.arrow_forwardWrite a Slater determinant for the lithide ion, Li.arrow_forward
- In your own words, explain why an object that has more symmetry elements is said to have higher symmetry than an object with fewer symmetry elements.arrow_forwardAssume that you are evaluating the integral of products of functions having symmetry labels in exercise 13.60, parts a-d. Which integrals, if any, are exactly zero due to symmetry considerations?arrow_forwardDetermine which single symmetry operation of the following point groups is equivalent to the given combination of multiple symmetry operations. a In C2v, C2v=? b In C2h, iC2=? c In D6h, C6h=? d In D2d, C2C2=? e In Oh, iS4=?arrow_forward
- 2. We can form a group of symmetry operations, with our operation being do one operation and do another. For a molecule with an isosceles triangle molecule like water, we talked about the 4 symmetry operations as E (do nothing), C2 (rotation by 180°), σv (the reflection of one hydrogen to the other), and σv’ (the reflection from front to back). By putting coordinates on each of the three atoms (z points along the C2 rotation axis, x points from one hydrogen to the other, and y points out of the plane of the molecule) find the “multiplication” table for this group.arrow_forwardWhat is the reducible representation for N205? Determine the reducible representation (T) that describes all the degrees of freedom .arrow_forwardUse symmetry properties to determine whether or not the integral ∫pxzpzdτ is necessarily zero in a molecule with symmetry D3h.arrow_forward
- Å L. Y → X 5 3 Consider the trimethylenecyclopropane molecule (point group D3h). Each carbon atom has a pz orbital to contribute to the pi- MOs of the molecule. Take these 6 pz orbitais as a basis set and answer the following questions: A) How do they divide into subsets of symmetry - equivalent orbitals? B) What representations are spanned by each subset? C) What are the 6 normalizedarrow_forwardHow to determine symmetry elements, σh and σν and how to differentiate between both. Also , how is σh related to E.arrow_forwardConsider the character table given below for the point group D3h, which includes molecules like cyclopropane and BF3. Suppose has been already determined a reducible representation for the collective motions of one such molecule, Γ(3N). What combination of irreps should we remove from it to account for translations & rotations, thereby yielding the purely vibrational representation, Γvib?arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

