
Write structural formulas for the following compounds.
- (a) C2H4Br2: δ 2.5 (d, 3H) and 5.9 (q, 1H)
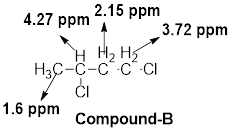
- (b) C4HgCl2: δ 1.60 (d, 3H), 2.15 (m, 2H), 3.72 (t, 2H), and 4.27 (m, 1H)
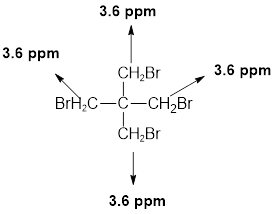
- (c) C5H8Br4: δ 3.6 (s, 8H)
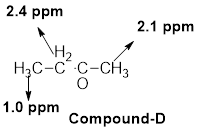
- (d) C4H8O: δ 1.0 (t, 3H), 2.1 (s, 3H), and 2.4 (quartet, 2H)
- (e) C4H8O2: δ 1.2 (t, 3H), 2.1 (s, 3H), and 4.1 (quartet, 2H); contains an ester
- (f) C4H8O2: δ 1.2 (t, 3H), 2.3 (quartet, 2H), and 3.6 (s, 3H); contains an ester
- (g) C4H9Br: δ 1.1 (d, 6H), 1.9 (m, 1H), and 3.4 (d, 2H)
- (h) C6H12O2: δ 1.5 (s, 9H) and 2.0 (s, 3H)
- (i) C7H14O: δ 0.9 (t, 6H), 1.6 (sextet, 4H), and 2.4 (t, 4H)
- (j) C5H10O2: δ 1.2 (d, 6H), 2.0 (s, 3H), and 5.0 (septet, 1H)
- (k) C5H11Br: δ 1.1 (s, 9H) and 3.2 (s, 2H)
- (l) C7H15Cl δ 1.1 (s, 9H) and 1.6 (s, 6H)
(a)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (a) is
There are 2 peaks observed in the given
A doublet is observed for 3 hydrogens at around
Another quartet peak for 1 hydrogen at around
Based on the above
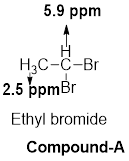
(b)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (b) is
A doublet is observed for 3 hydrogens at around
A multiplet is observed for 2 hydrogens at around
One triplet peak is observed for 2 hydrogens at around
Another multiplet is observed for 1 hydrogen at around
Thus, the structure of the compound (b) is 1,3-dichlorobutane:
(c)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (c) is
This molecule only one singlet is observed for 8 hydrogens at around
Therefore, the structure of the compound (c) is given below and this molecule symmetrical.
(d)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (d) is
A triplet is observed for 3 hydrogens at around
One singlet is observed for 3 hydrogens at around
Another quartet is observed for 2 hydrogens at around
Thus, the structure of the compound (d) is,
(e)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (e) is
A triplet is observed for 3 hydrogens at around
One singlet is observed for 3 hydrogens at around
Another quartet is observed for 2 hydrogens at around
Thus, the structure of the compound (e) is,
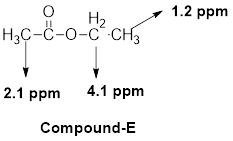
(f)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (f) is
A triplet is observed for 3 hydrogens at around
One singlet is observed for 3 hydrogens at around
Another quartet is observed for 2 hydrogens at around
Thus, the structure of the compound (f) is,
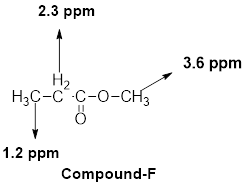
(g)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (g) is
A doublet is observed for 6 hydrogens at around
One multiplet is observed for 1 hydrogen at around
Another multiplet is observed for 2 hydrogens at around
Thus, the structure of the compound (g) is,
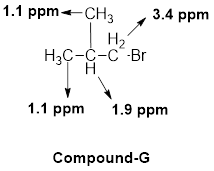
(h)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (h) is
A singlet is observed for 9 hydrogens at around
Another one singlet is observed for 3 hydrogens at around
Thus, the structure of the compound (h) is,
(i)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (i) is
A singlet is observed for 6 hydrogens at around
One sextet is observed for 4 hydrogens at around
Another triplet peak for 4 hydrogens at around
Thus, the structure of the compound (i) is,
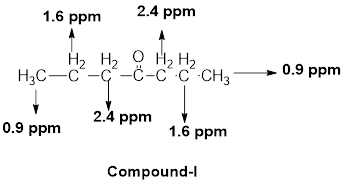
(j)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (J) is
A doublet is observed for 6 hydrogens at around
One septet is observed for 1 hydrogen at around
Another singlet peak for 3 hydrogens at around
Thus, the structure of the compound (J) is,
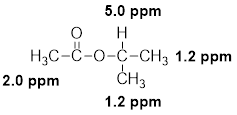
(k)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (K) is
A singlet is observed for 9 hydrogens at around
One singlet is observed for 2 hydrogens at around
Thus, the structure of the compound (K) is,
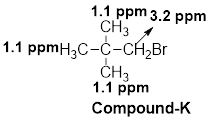
(l)
Interpretation:
The structural formula of the given compound has to be proposed with the help of given molecular formula and its
Concept introduction:
The
Chemical shift: The NMR spectrum of any compound is taken with reference to a standard compound called reference compound. Generally, tetramethylsilane (TMS) is taken as the reference compound. The methyl protons of TMS are equivalent and produces only one sharp peak at the rightmost end of the scale.
The distance between the TMS signal and the signals produced by the compound is called the chemical shift. Chemical shift basically measures the shift in the signal position of the compound with respect to the reference signal.
Chemical shift in delta scale is given as,
Explanation of Solution
The given molecular formula (L) is
A singlet is observed for 9 hydrogens at around
One singlet is observed for 6 hydrogens at around
Thus, the structure of the compound (L) is,
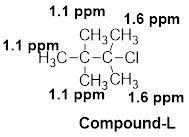
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry, Loose-leaf Version
Additional Science Textbook Solutions
General, Organic, & Biological Chemistry
Elementary Principles of Chemical Processes, Binder Ready Version
General Chemistry: Atoms First
Chemistry: Atoms First
Basic Chemistry (5th Edition)
- 2. Calculate the enthalpy change for the reaction : C4H4 (9) + H2 (9) C2H6 (g) Given the following data: i) C2H4 (9) + 3 O2 (9) → 2 CO2 (9) + 2 H2O (1) AH° = - 1410.9 kJ 2 C2H6 (9) + 7 02 (9) →4 CO2 (9) + 6 H20 (1) ii) iii) 2 H2 (9) + O2 (g) 2 H2O (1) AH° = - 3119.4 kJ AH° = - 571.6 kJ 3. Calculate the standard enthalpy change for the reaction, C2H2 (g) + 2 H2 (9g) - C2H6 (9) Given that: i) 2 C2H2 (9) + 5 02 (g) 4 CO2 (9) + 2 H2O (1) AH° = - 2599 kJ 3119 kJ ii) 2 C2H6 (9) + 7 02 (9) - 4 CO2 (9) + 6 H20 (1) AH° = iii) 2 H2 (9) + O2 (9) → 2 H2O () AH° = - 572 kJ The enthalpy of combustion of methane is -891 kJmol-1. Calculate the enthalpy of formation of methane if the enthalpy of formation of carbon dioxide and water are - 394 kJmol-1 and -286 kJmol respectively. Chemical equation for formation of 4. methane: C(s) + 2H2(9) CH4(9) Given: AH = -891 kJmol-1 CH4(9) + 202(9) 2H2O() + CO2(g) C(s) + Oz(g) → CO2(9) H2(g) + 2 O2 - H20 AH = -394 kJmol-1 AH = -286 kJmol-1arrow_forward(1) C(s) + O2(g) CO2(8) AH, = -393.5 kJ AH = -283.0 kJ Using these data, calculate the enthalpy for the combustion of C to CO: (2) CO(g) + 02(8) CO2(8) - %3D (3) C(s) + 02(8) CO(g) AH3 = ? 24 - O A) 403.5 O B) 676.5 403.5 O D) 110.5 O E) -110.5arrow_forwardFind the AHn for the following reaction: B2H6 (g) + 6 Cl2(9) → 2 BC13(g) + 6 HC(9) given the following reactions and subsequent AH° values $$ H3 BO3 (g) + 3 HC((g) ΔΗ'- -113.1 kJ BC13 (g) + 3 H20(1) → B2 H6 + 6 H2O(1) → 2 H3 BO3(9) + 6 H2 (9) AH° = -496 kJ 1 HC(g) AH° = -95.5 kJ (6)1 + (6)H? $$arrow_forward
- Construct the expression for Kc for the following reaction. C(s) + H,O(g) = CO(g) + H,(g)arrow_forwardP2C.4 From the following data, determine A,H° for diborane, B,H,(g), at 298 K: (1) B,H,(g) + 30,(g) → B,O,(s) +3 H,O(g) A̟H° =-1941kJ mol -> %3D (2) 2B(s) + 를0,(g) → B,O, (s) A̟H® =-2368 kJ mol (3) H,(g)+±O,(g) → H,O(g) A,H° =-241.8kJ mol %3Darrow_forwardCalculate Δ Hrxn for Ca(s) + 1/2O2(g) + CO2(g) -> CaCO3(s) Given the following set of reactions: Ca(s) + 1/2O2(G) -> CaO(2) ΔH= -635.1kJ CaCO3(s) -> CaO(s) + CO2(g) ΔH = 178.3kJ a.) 813.41 kJ b.) -813.4 kJ c.) 456.8 kJ d.) -456.8 kJarrow_forward
- Find the deltaH for the reaction below, given the following reactions and subsequent deltaH values: N2H4(l) + H2(g) —> 2NH3 (g) N2H4(l) + CH4O(l) —> CH2O (g) + N2(g) + 3H2(g)—— deltaH= -32.8kj N2(g)+ 3H2(g) —> 2NH3 (g)—deltaH=-57.2 kj CH4O(l) —>CH2O(g) + H2(g) —-deltaH= -65 kj SIG FIGSarrow_forwardOf the following reactions or processes, is there one for which the enthalpy change is zero?(A). Hydrogen formation reaction at 1 atm and 25°C. (B). Transformation of one ethane rotamer into another. (C). Dissolution of O2 in water. (D). All three processes have non-zero enthalpy changes associated with them.arrow_forwardUsing the provided bond energies, estimate the AHrxn for the generic reaction below. The skeletal structures of each molecule are shown below the balanced equation, where each (-) represents a single bond. 2 A2 B → 2 A2 + B2 2 (A – B – A) → 2 (A – A) + (B – B) - - Bond energies: (A-B) = 150. kJ/mol; (A-A) = 200. kJ/mol; (B-B) %3D = 250. kJ/mol O -300. kJ O 350. kJ O -50. kJ 600. kJ O 1250. kJarrow_forward
- The enthalpy changes accompanying the dissociation of successive bonds in NH3(g) are 460, 390, and 314 kJ mol-1 , respectively. (a) What is the mean enthalpy of an N-H bond? (b) Do you expect the mean bond internal energy to be larger or smaller than the mean bond enthalpy? (c) Use these values,along with tabulated bond enthalpies for the N-N and H-H bonds to estimate the standard enthalpy of formation of gaseous ammonia, NH3 , at 298 K.arrow_forward(b) An aqueous solution of acetic acid can be prepared by the reaction of ethanol with oxygen : CH3CH2OH (1) + O2 (g) –→ CH3COOH (1) + H2O (1) Write thermochemical equations for enthalpy of formation of the given compounds and calculate AH for the reaction, given the following data. Enthalpy of formation, AHf (kJmol") -277.8 Compound CH;CH2OH (1) CH3COOH (I) H2O (1) -485.0 -286.0arrow_forward(d) If the mass of the metal is 196 grams, what is its specific heat capacity?(e) What is the change of water’s entropy? Note the volume of water stays the same.(f) What is the change of total entropy of the system, including both water and metal?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





