
Concept explainers
(a)
Interpretation:
The missing reactant in the given conversion has to be identified.
Concept Introduction:
Alkylation:
Benzene undergoes alkylation reaction in presence of

Halogenation:
Benzene undergoes halogenation reaction in presence of
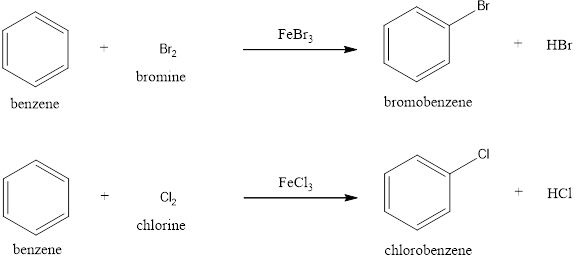
(c)
Interpretation:
The missing reactant in the given conversion has to be identified.
Concept Introduction:
Aromatic hydrocarbons undergo substitution reactions rather than addition reactions. Even though the aromatic hydrocarbon has double bonds in its structure, they undergo substitution reaction only because the double bonds are involved in delocalized bonding that is present in the ring system.
Alkylation:
Benzene undergoes alkylation reaction in presence of

Halogenation:
Benzene undergoes halogenation reaction in presence of

(c)
Interpretation:
The missing product in the given conversion has to be identified.
Concept Introduction:
Aromatic hydrocarbons undergo substitution reactions rather than addition reactions. Even though the aromatic hydrocarbon has double bonds in its structure, they undergo substitution reaction only because the double bonds are involved in delocalized bonding that is present in the ring system.
Alkylation:
Benzene undergoes alkylation reaction in presence of

Halogenation:
Benzene undergoes halogenation reaction in presence of

Trending nowThis is a popular solution!

Chapter 13 Solutions
General, Organic, And Biological Chemistry
- Given the reaction: benzoic anhydride + methanol Draw the structure of the tetrahedral intermediate that is formed and indicate how it dissociates to give the reaction product (use arrows).arrow_forwardWrite the chemical formulas for the products formed when each of the following hydrocarbons undergoes complete combustion. a. CH4 b. C4H10 c. C4H6 d. C6H6arrow_forwardAlcohols are very useful starting materials for the production of many different compounds. The following conversions, starting with 1-butanol, can be carried out in two or more steps. Show the steps (reactants/catalysts) you would follow to carry out the conversions, drawing the formula for the organic product in each step. For each step, a major product must be produced. (See Exercise 62.) (Hint: In the presence of H+, an alcohol is converted into an alkene and water. This is the exact reverse of the reaction of adding water to an alkene to form an alcohol.) a. 1-butanol butane b. 1-butanol 2-butanonearrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





