
(a)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as Lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 41E
Electron dot structure and structural formula of
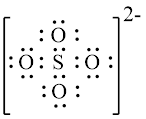
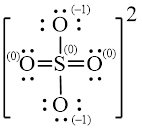
Explanation of Solution
In molecule,
Figure 1
Figure 2
Solid line, in Figure 2, between the atoms is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the atoms present in that bond.
An electron dot structure and structural formula of
(b)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as Lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 41E
Electron dot structure and structural formula of
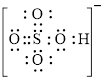
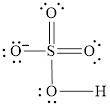
Explanation of Solution
In molecule
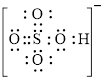
Figure 3

Figure 4
Each solid line, in Figure 4, between the atoms is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the atoms present in that bond.
An electron dot structure and structural formula of
(c)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as Lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 41E
Electron dot structure and structural formula of
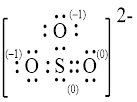
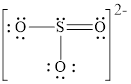
Explanation of Solution
In molecule
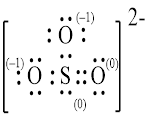
Figure 5

Figure 6
Each solid line, in Figure 6, between the atoms is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the atoms present in that bond.
An electron dot structure and structural formula of
(d)
Interpretation:
The electron dot formula and structural formula of
Concept introduction:
An electron dot formula is a way of representing the molecular structure in which electrons are represented by a dot. Structural formula is a way in which atoms are linked together through a solid line. This solid line represents the covalent bond. An electron dot structure is known as Lewis structure. Electron dot structure indicates the valence electrons of an atom which are involved in bonding.
Answer to Problem 41E
Electron dot structure and structural formula of
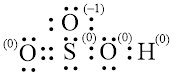
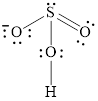
Explanation of Solution
In molecule
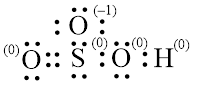
Figure 7

Figure 8
Each solid line, in Figure 8, between the atoms is the covalent bond which is made up of two electrons. This bond is formed by sharing of electrons between the atoms present in that bond.
An electron dot structure and structural formula of
Want to see more full solutions like this?
Chapter 12 Solutions
Introductory Chemistry: Concepts and Critical Thinking (8th Edition)
- 2-98 Explain how the ionization energy of atoms changes when proceeding down a group of the Periodic Table and explain why this change occurs.arrow_forwardWhy do nonmetals tend to form anions rather than cations?arrow_forward3-58 In Section 2-3B, we saw that there are seven diatomic elements. (a) Draw Lewis structures for each of these diatomic elements. (b) Which diatomic elements are gases at room temperature? Which are liquids? Which are solids?arrow_forward
- If X is in the period 5, the ion formed has the same electron configuration as the noble gasarrow_forwardSuppose an element from group IIIA(13), and period 3 of the periodic table forms an ionic compound with the element that has an electronic configuration of 1s2 2s2 2p5. What would be the formula of the compound, and what would be the name of the compound?arrow_forwardHow many electrons will beryllium need to be removed in order for it to become a stable ion?arrow_forward
- What is the relationship between the tendency of a main-group element to form a monatomic ion and its position in the periodic table? In what part of the table are the main-group ele-ments that typically form cations? Anions?arrow_forwardwrite the lewis structure of the molecule including the resonance forms, and the formal charges. the relative positions of the atoms are given as references answer #3arrow_forwardIn the nitric oxide molecule (NO), which atom is expected to hold the partial negative charge? A. the N atom B. the O atom C. neither atom holds a partial negative chargearrow_forward
- Which one of the following does not occur as diatomic molecules in elemental form? Group of answer choices oxygen hydrogen sulfur nitrogen brominearrow_forwardstructural formula for the nitrite ion, NO2–arrow_forwardBarium ions carry a 2+ charge, and nitrogen ions carry a 3- charge. What is the chemical formula for the iconic compound barium nitride?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


