
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12, Problem 12.4P
Problems 12.3 through 12.8 refer to the Pxy diagram for ethanol(1)/ethyl acetate(2) at 70°C shown in Fig. 12.19.
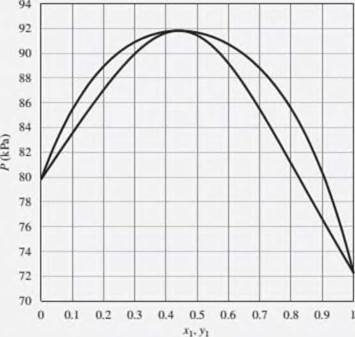
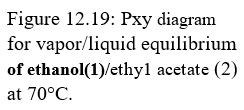
12.4. The pressure above a mixture of ethanol and ethyl acetate at 70°C is measured to be 78 kPa. What are the possible compositions of the liquid and vapor phases?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. Liquid heptane is stored in a 100,000-L storage vessel that is vented directly to air. The
heptane is stored at 25°C and 1 atm pressure. The liquid is drained from the storage
vessel and all that remains in the vessel is the air saturated with heptane vapor.
a. Is the vapor in the storage vessel flammable?
b. What is the TNT equivalent for the vapor remaining in the vessel?
c. If the vapor explodes, what is the overpressure 50 m from the vessel?
d. What damage can be expected at 50 m?
2. You have decided to use a vacuum purging technique to purge oxygen from a reactor
vessel to reduce the concentration to 2.0% (mol). The reactor is 18 ft diameter and 40 ft
tall. The temperature is 80°F. Assume that the vacuum purge goes from atmospheric
pressure to 10.0 psia. How many purge cycles are required and how many total moles of
nitrogen must be used? Assume the purge is done with pure nitrogen.
3. If the purging described in problem 2 takes place using nitrogen that has 1% (mol)
oxygen in it, how many vacuum purge cycles are required? How many total moles of the
inert gas must be used?
4. If the purging described in problem 2 is done by way of a "sweep-through" purge instead
of a vacuum purge, for how long (in minutes) must the inert gas flow through the vessel
if there is a 20 psig supply of pure nitrogen available at 150 CFM (ft³/min)? How much
nitrogen must be used (lbm)?
5. Look at Figure 7-14. Determine the voltage developed between the steel nozzle and the
grounded vessel, and how much energy is stored in the nozzle. Explain the potential
hazards for cases A and B from the following table:
Case A
Case B
Hose length (ft)
75
75
Hose diameter (in)
2.0
2.0
Flow rate (gpm)
30
30
Liquid conductivity (mho/cm)
2x10-8
1x10-14
Dielectric constant
2.3
25
Density (g/cm³)
0.8
0.9
6. In Problem 5, case B, what would be the most effective way to reduce the potential
hazards in this situation?
Chapter 12 Solutions
Introduction to Chemical Engineering Thermodynamics
Ch. 12 - Prob. 12.1PCh. 12 - Prob. 12.2PCh. 12 - Prob. 12.3PCh. 12 - Problems 12.3 through 12.8 refer to the Pxy...Ch. 12 - Problems 12.3 through 12.8 refer to the Pxy...Ch. 12 - Problems 12.3 through 12.8 refer to the Pxy...Ch. 12 - Problems 12.3 through 12.8 refer to the Pxy...Ch. 12 - Problems 12.3 through 12.8 refer to the Pxy...Ch. 12 - Problems 12.9 through 12.14 refer to the Txy...Ch. 12 - Problems 12.9 through 12.14 refer to the Txy...
Ch. 12 - Problems 12.9 through 12.14 refer to the Txy...Ch. 12 - Problems 12.9 through 12.14 refer to the Txy...Ch. 12 - Problems 12.9 through 12.14 refer to the Txy...Ch. 12 - Problems 12.9 through 12.14 refer to the Txy...Ch. 12 - Prob. 12.15PCh. 12 - Problems 12.16 through 12.21 refer to the Pxy...Ch. 12 - Problems 12.16 through 12.21 refer to the Pxy...Ch. 12 - Problems 12.16 through 12.21 refer to the Pxy...Ch. 12 - Problems 12.16 through 12.21 refer to the Pxy...Ch. 12 - Problems 12.16 through 12.21 refer to the Pxy...Ch. 12 - Prob. 12.21PCh. 12 - Problems 12.22 through 12.28 refer to the Txy...Ch. 12 - Problems 12.22 through 12.28 refer to the Txy...Ch. 12 - Problems 12.22 through 12.28 refer to the Txy...Ch. 12 - Problems 12.22 through 12.28 refer to the Txy...Ch. 12 - Problems 12.22 through 12.28 refer to the Txy...Ch. 12 - Problems 12.22 through 12.28 refer to the Txy...Ch. 12 - Prob. 12.28PCh. 12 - Problems 12.29 through 12.33 refer to the xy...Ch. 12 - Problems 12.29 through 12.33 refer to the xy...Ch. 12 - Problems 12.29 through 12.33 refer to the xy...Ch. 12 - Problems 12.29 through 12.33 refer to the xy...Ch. 12 - Problems 12.29 through 12.33 refer to the xy...Ch. 12 - Consider a binary liquid mixture for which the...Ch. 12 - Prob. 12.35PCh. 12 - Prob. 12.36P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Similar questions
- 2. You have decided to use a vacuum purging technique to purge oxygen from a reactor vessel to reduce the concentration to 2.0% (mol). The reactor is 18 ft diameter and 40 ft tall. The temperature is 80°F. Assume that the vacuum purge goes from atmospheric pressure to 10.0 psia. How many purge cycles are required and how many total moles of nitrogen must be used? Assume the purge is done with pure nitrogen.arrow_forwardAn 8-foot ion exchange bed needs to be backwashed with water to remove impurities. The particles have a density of 1.24 g/cm³ and an average size of 1.1 mm. Calculate: a. The minimum fluidization velocity using water at 30°C? b. The velocity required to expand the bed by 30%? Assumptions: The ion exchange bed particles are spherical (sphericity = 1.1), and the minimum fluidization porosity (ɛM) is 0.3. Notes: At 30°C, the viscosity (μ) of water is 0.797 cP, and the density (ρ) is 0.995 g/cm³.arrow_forwardfluidized bed reactor uses a solid catalyst with a particle diameter of 0.25 mm, a bulk density of 1.50 g/mL, and a sphericity of 0.90. Under packed bed conditions, the porosity is 0.35, and the bed height is 2 m. The gas enters from the bottom of the reactor at a temperature of 600°C, with a viscosity of 0.025 cP and a density of 0.22 lb/ft³. At minimum fluidization, the porosity reaches 0.45. Calculate: a. The minimum superficial velocity (VM) of the gas entering the fluidized column. b. The bed height if V = 2 VM c. The pressure drop under conditions where V =2.5 VMarrow_forward
- A fluidized bed reactor uses a solid catalyst with a particle diameter of 0.25 mm, a bulk density of 1.50 g/mL, and a sphericity of 0.90. Under packed bed conditions, the porosity is 0.35, and the bed height is 2 m. The gas enters from the bottom of the reactor at a temperature of 600°C, with a viscosity of 0.025 cP and a density of 0.22 lb/ft³. At minimum fluidization, the porosity reaches 0.45. Calculate: a. The minimum superficial velocity (VM) of the gas entering the fluidized column. b. The bed height if V = 2 VM c. The pressure drop under conditions where V =2.5 VMarrow_forwardPlease answer 5.8arrow_forwardPlease answer 5.6arrow_forward
- You have been tasked with figuring out how to suppress changes in the supply flow rate to a reactorfor which it is desired to keep the inlet flow rate as constant as possible. You are considering designing a surgetank to place upstream of the reactor and then installing a pump on the line between that surge tank and thereactor. A surge tank is one with a weir inside it, which is a partial wall separating the tank volume into twoconnected sections allowing for flow under the weir between the two sections. The variable inlet mass flow,wi(t) flows into volume 1 and then flows due to hydrostatic pressure at a mass flow rate of w1(t) into volume 2.The weir causes a flow resistance, R1, such that w1 = (h1-h2)/R1. Fluid is then pumped out of volume 2 at thedesired constant mass flow rate of w2. Make a summary table of the three transfer functions written in standard form and their keyparameters (gains, time constants) in terms of the physical system parameters (A1, A2, , A, R…). Checkif/how…arrow_forwardThe vapor pressure of Toluene at 50°C in Pa? Find it on perry's chemical engineering handbook 9th or 8th editionarrow_forwardHydrogen (H₂) is considered a clean energy carrier. For its use as a fuel, hydrogen is stored at 5 bar insidea cylindrical tank made of nickel (Ni) with 7 cm inner diameter, 1.2 mm thickness, and the length of L. Thetank is maintained at 358 K. Unfortunately, a small amount of hydrogen diffuses out of the tank, slowlydepleting its contents. You may assume that the hydrogen pressure outside the tank is essentially zero andconvective resistance inside and outside of the cylinder is negligible.• Solubility of H2 in Ni at 358 K = 0.00901 kmol/m3·bar• DH2, Ni at 358 K = 1.2 x 10-12 m2/sCalculate the maximum length of the nickel tank wall to ensure that the hydrogen loss does not exceed0.01 kg per year.arrow_forward
- You just took out a cold soda can (at 1 oC) from the refrigerator. Calculate thetemperature of the soda can after the can is placed in a room (at 31 oC, h = 100 W/m2-K) for 60 mins (we all know that soda tastes much better when it is cold!). • k = 0.617 W/m-K, density = 996 kg/m3, Cp = 4178 J/Kg-K• Height = 10 cm & Diameter = 5 cmCalculate the temperature of the soda can surface at the middle point of the heightusing 2-D analysis.arrow_forwardA thick nickel wall is exposed to pure 5 bar H2(g) at 85 oC on one side of its surface (13 pts).(a) Assuming thermodynamic gas-solid equilibrium, calculate the H2 concentration at the surface ofthe nickel wall. (b) Assuming that the concentration of H2 at the surface is constant, determine the concentration ofH2 at the penetration depth in percentage of its concentration at the wall surfacearrow_forwardCan you provide me the answer of these pleasearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY
Chemical Equilibrium Constant K - Ice Tables - Kp and Kc; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=J4WJCYpTYj8;License: Standard Youtube License