
Concept explainers
(a)
Interpretation:
The mass spectrum fragmentation of ethyl bromide at m/z=110(98%) is to be stated.
Concept introduction:
In mass spectroscopy, compounds can be identified on the basis of mass of compound. When the compound breaks into fragment then they can be distinguished from the other compounds. This technique is also used to differentiate the isotopes of compounds. This technique did not interact with
Answer to Problem 12.42AP
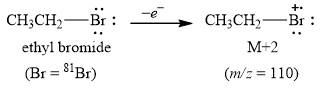
The peak at m/z=110(98%) is observed due to bromide isotope [81Br] that is [M+2].
Explanation of Solution
The molecule ethyl bromide contains carbon, hydrogen, bromide atoms. In this molecule carbon and hydrogen mainly exist in one isotope form 12C with

Figure 1
The peak at m/z=110(98%) is observed due to [81Br] that is [M+2].
The mass spectrum fragmentation of ethyl bromide at m/z=110(98%) is due to [81Br] that is [M+2] as shown in Figure 1.
(b)
Interpretation:
The mass spectrum fragmentation of ethyl bromide at m/z=108(100%) is to be stated.
Concept introduction:
In mass spectroscopy, compounds can be identified on the basis of mass of compound. When the compound breaks into fragment then they can be distinguished from the other compounds. This technique is also used to differentiate the isotopes of compounds. This technique did not interact with electromagnetic radiation. Two peaks are used to identify the compound, first, the molecular ion peak which is the mass of the compound and second, the base peak which is the most abundant element peak. It may be same or different.
Answer to Problem 12.42AP
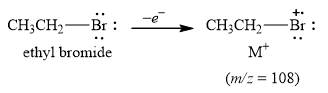
The peak at m/z=108(100%) is observed due to [79Br] that is [M] the molecular ion peak.
Explanation of Solution
The mass of the compound, mass=108, gives the molecular ion peak when it gives away one electron. The peak at m/z=108(100%) is due to [79Br] as shown below.

Figure 2
The mass spectrum fragmentation of ethyl bromide at m/z=108(100%) is due to [79Br] molecular ion peak as shown in Figure 2.
(c)
Interpretation:
The mass spectrum fragmentation of ethyl bromide at m/z=81(5%) is to be stated.
Concept introduction:
In mass spectroscopy, compounds can be identified on the basis of mass of compound. When the compound breaks into fragment then they can be distinguished from the other compounds. This technique is also used to differentiate the isotopes of compounds. This technique did not interact with electromagnetic radiation. Two peaks are used to identify the compound, first, the molecular ion peak which is the mass of the compound and second, the base peak which is the most abundant element peak. It may be same or different.
Answer to Problem 12.42AP
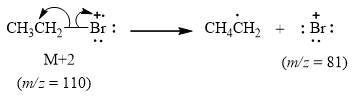
The peak at m/z=81(5%) is due to bromonium cation of [81Br].
Explanation of Solution
When the ethyl bromide [M+2] loses ethyl radical it release bromonium cation [Br+] whose mass=81. This gives peak at m/z=81(5%) when [81Br] is present. It is shown below.

Figure 3
The mass spectrum fragmentation of ethyl bromide at m/z=81(5%) is due to bromonium cation [Br+] of [81Br] isotope as shown in Figure 3.
(d)
Interpretation:
The mass spectrum fragmentation of ethyl bromide at m/z=79(5%) is to be stated.
Concept introduction:
In mass spectroscopy, compounds can be identified on the basis of mass of compound. When the compound breaks into fragment then they can be distinguished from the other compounds. This technique is also used to differentiate the isotopes of compounds. This technique did not interact with electromagnetic radiation. Two peaks are used to identify the compound, first, the molecular ion peak which is the mass of the compound and second, the base peak which is the most abundant element peak. It may be same or different.
Answer to Problem 12.42AP
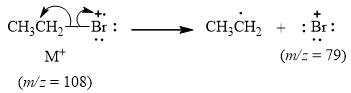
The peak at m/z=79(5%) is due to bromonium cation of [79Br].
Explanation of Solution
When the ethyl bromide [M] loses ethyl radical it release bromonium cation [Br+] with mass=79. This gives peak at m/z=79(5%) when [79Br] is present. It is shown below.

Figure 4
The mass spectrum fragmentation of ethyl bromide at m/z=79(5%) is due to bromonium cation [Br+] of [79Br] isotope as shown in Figure 4.
(e)
Interpretation:
The mass spectrum fragmentation of ethyl bromide at m/z=29(61%) is to be stated.
Concept introduction:
In mass spectroscopy, compounds can be identified on the basis of mass of compound. When the compound breaks into fragment then they can be distinguished from the other compounds. This technique is also used to differentiate the isotopes of compounds. This technique did not interact with electromagnetic radiation. Two peaks are used to identify the compound, first, the molecular ion peak which is the mass of the compound and second, the base peak which is the most abundant element peak. It may be same or different.
Answer to Problem 12.42AP
The peak at m/z=29(61%) is due to ethyl cation [C2H5+]
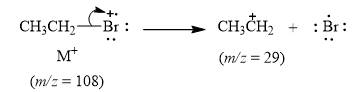
Explanation of Solution
When the ethyl bromide breaks into fragment, it releases ethyl cation [C2H5+] with mass=29. Therefore, peak at m/z=29(61%) is observed. It is shown below.

Figure 5
The mass spectrum fragmentation of ethyl bromide at m/z=29(61%) is due to ethyl cation [C2H5+] as shown in Figure 5.
(f)
Interpretation:
The mass spectrum fragmentation of ethyl bromide at m/z=28(25%) is to be stated.
Concept introduction:
In mass spectroscopy, compounds can be identified on the basis of mass of compound. When the compound breaks into fragment then they can be distinguished from the other compounds. This technique is also used to differentiate the isotopes of compounds. This technique did not interact with electromagnetic radiation. Two peaks are used to identify the compound, first, the molecular ion peak which is the mass of the compound and second, the base peak which is the most abundant element peak. It may be same or different.
Answer to Problem 12.42AP
The peak at m/z=28(25%) is due to ethyl radical cation [C2H4•+].
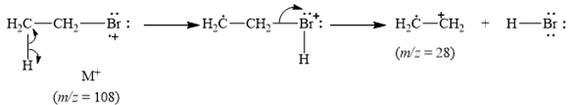
Explanation of Solution
When the ethyl bromide breaks into fragment, it releases ethyl radical cation [C2H4•+] with mass=28. Therefore, peak at m/z=28(25%) is observed. It is shown below.

Figure 6
The mass spectrum fragmentation of ethyl bromide at m/z=28(25%) is due to ethyl radical cation [C2H4•+] as shown below in Figure 6.
(g)
Interpretation:
The mass spectrum fragmentation of ethyl bromide at m/z=27(53%) is to be stated.
Concept introduction:
In mass spectroscopy, compounds can be identified on the basis of mass of compound. When the compound breaks into fragment then they can be distinguished from the other compounds. This technique is also used to differentiate the isotopes of compounds. This technique did not interact with electromagnetic radiation. Two peaks are used to identify the compound, first, the molecular ion peak which is the mass of the compound and second, the base peak which is the most abundant element peak. It may be same or different.
Answer to Problem 12.42AP
The peak at m/z=28(25%) is due to ethene cation [CH=CH+].
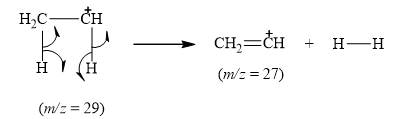
Explanation of Solution
When the ethyl bromide breaks into fragment, it releases ethene cation [CH=CH+] with mass=27. Therefore, peak at m/z=27(53%) is observed. It is shown below.

Figure 7
The mass spectrum fragmentation of ethyl bromide at m/z=27(53%) is due to ethene cation [CH=CH+] as shown in Figure 7.
Want to see more full solutions like this?
Chapter 12 Solutions
Organic Chemistry
- 4. Which one of the following is trans-1-tert-butyl-3-methylcyclohexane in its most stable conformation? (NOTE: Correct answer must be trans- and must have a 1,3-arrangement of groups.) C(CH3)3 CH₁₂ A H,C D H₂C C(CH) C(CH3)3 C B CH C(CH) C(CH3)3 Earrow_forwardPredict the Product. Predict the major organic product for the following reaction:arrow_forwardNonearrow_forward
- 3. Which one of the following is the lowest energy, most stable conformation of 1-bromopropane? H H H H H H H H CH3 HH Br H CH3 b b b b b CH3 A Br Br H H B CH3 Br H C H H H D CH3 H Br H E Harrow_forwardIn evolution, migration refers to the movement of alleles between populations. In your drawings, compare and contrast migration in evolutionary terms vs. in ecological terms. True Falsearrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. Problem 31 I 1 :0: O: C 1 1 H Na Select to Add Arrows CH3CH2CCNa 1arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





