
Concept explainers
Draw the organic products formed when allylic alcoholA is treated with each reagent.
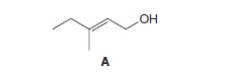
a.
b.
c.
d.
e.
f.
g. [1]
h.
(a)
Interpretation: The product formed when A is treated with
Concept introduction: The addition of
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
When A is treated with
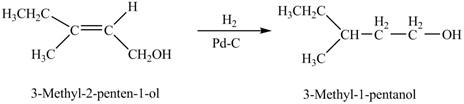
Figure 1
The product formed when A is treated with
(b)
Interpretation: The product formed when A is treated with
Concept introduction: In presence of peroxide alkene is oxidized to epoxide this is known as epoxidation. The weak pi bond of alkene and weak
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
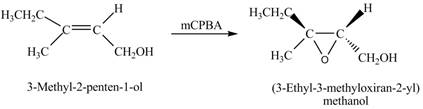
Figure 2
The product formed when A is treated with
(c)
Interpretation: The product formed when A is treated with
Concept introduction: Alcohols are oxidized to different carbonyl compounds depending upon the reagents and alcohol used. In presence of strong oxidizing reagents such as
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
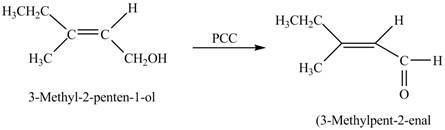
Figure 3
The product formed when A is treated with
(d)
Interpretation: The product formed when A is treated with
Concept introduction: Alcohols are oxidized to different carbonyl compounds depending upon the reagents and alcohol used. In the presence of strong oxidizing reagents such as
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
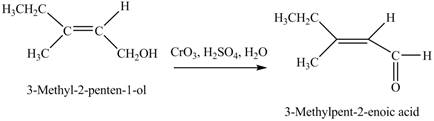
Figure 4
The product formed when A is treated with
(e)
Interpretation: The product formed when A is treated with Sharpless reagent
Concept introduction: Sharpless epoxidation involves the oxidation of double bond between carbon atoms to epoxide. This oxidation occurs only in allylic alcohol. This is an enantioselective oxidation, which means predominantly one enantiomer is formed. Sharpless reagents are
Answer to Problem 12.39P
The product formed when A is treated with Sharpless reagent
Explanation of Solution
There are two different chiral diethyl tartrate isomers,
When epoxidation is done using
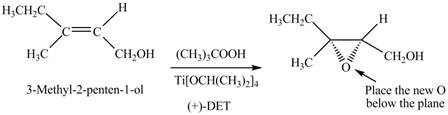
Figure 5
The product formed when A is treated with Sharpless reagent
(f)
Interpretation: The product formed when A is treated with Sharpless reagent
Concept introduction: Sharpless epoxidation involves the oxidation of double bond between carbon atoms to epoxide. This oxidation occurs only in allylic alcohol. This is an enantioselective oxidation, which means predominantly one enantiomer is formed. Sharpless reagents are
Answer to Problem 12.39P
The product of formed when A is treated with Sharpless reagent
Explanation of Solution
There are two different chiral diethyl tartrate isomers,
When epoxidation is done using
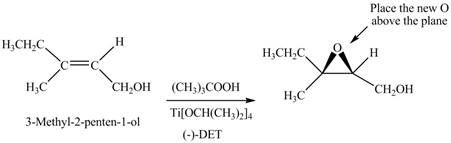
Figure 6
The product formed when A is treated with Sharpless reagent
(g)
Interpretation: The product formed when A is treated with given reagent is shown in Figure 7.
Concept introduction: Alcohols on treatment with phosphorus tribromide gives alkyl bromide. The
Answer to Problem 12.39P
The product formed in the given reaction is shown in Figure 7.
Explanation of Solution
The given reaction is,
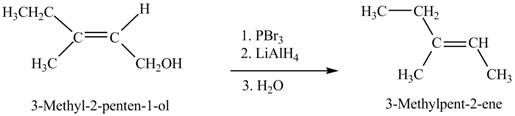
Figure 7
When the given alcohol is treated with
The product formed when A is treated with given reagent is shown in Figure 7.
(h)
Interpretation: The product formed when A is treated with
Concept introduction:
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
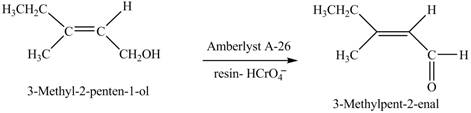
Figure 8
The product formed when A is treated with
Want to see more full solutions like this?
Chapter 12 Solutions
PKG ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry: Matter and Change
Chemistry
Chemistry & Chemical Reactivity
Chemistry (7th Edition)
Thermodynamics, Statistical Thermodynamics, & Kinetics
- What is the major organic product obtained from the following reaction? A. CI B. H 1. (CH3CH₂)2CuLi 2. H3O+ C. D. •ay.a OHarrow_forwardWhat is the major organic product obtained from the following reaction? a. 1 b.2 c. 3 d. 4 -CC-H 1. BH3 2. H₂O₂, NaOH 1 3 OH 2 4 CH₂arrow_forwardDraw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH c.HCl + ZnCl2 d.HBr e.SOCl2, pyridine f.PBr3 g.TsCl, pyridine h. [1] NaH; [2] CH3CH2Br [1] i.TsCl, pyridine; [2] NaSH j.POCl3, pyridinearrow_forward
- B. Draw structures for the products in the following reactions. my NH₂ NH₂ 1. LIAIH4 2. H₂O* xs EtBr 1. Mel 2. Ag₂O, H,O, heat acetone, formaldehyde cat. HCIarrow_forwardDraw the product of the following reaction sequence. CI 1. Mg(s), THF 2. CO₂(s) 3. H₂O+arrow_forwardWhat is the major organic product obtained from the following reaction? CH3CH₂CH₂CH₂C=CH a. 2 b. 3 c. 1 O d. 4 H₂O HgSO4 H₂SO4 OH OH 3. OH OH 2 4arrow_forward
- Ozonolysis is the application of reaction O A. substitution B. oxidation O C. addition O D. eliminationarrow_forwardtaken in order to gor the product Please explain the mechanisms/stepsarrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
