
a)
Interpretation:
The expected product between the
Answer to Problem 21VC
The ethyl chloride undergoes SN2 reaction with Na+SCH3- and NaOH to yield a product.
The product is CH3CH2SCH3, CH3CH2OH.
Here the leaving group is Cl.
The nucleophile is SCH3-,OH.
Hence it undergoes SN2 reaction.
Explanation of Solution
The ethyl chloride undergoes SN2 reaction with Na+SCH3- and NaOH to yield a product.
The product is CH3CH2SCH3, CH3CH2OH.
Here the leaving group is Cl.
The nucleophile is SCH3-,OH.
Hence it undergoes SN2 reaction.
The ethyl chloride undergoes SN2 reaction with Na+SCH3- and NaOH to yield a product.
The product is CH3CH2SCH3, CH3CH2OH.
Here the leaving group is Cl.
The nucleophile is SCH3-,OH.
Hence it undergoes SN2 reaction.
b)
Interpretation:
The expected product between the alkyl halide CH3CH2Cl+Na+SCH3- and NaOH is interpreted.
Answer to Problem 21VC
The alkyl halide undergoes SN1 reaction with Na+SCH3- to yield a SCH3 Substituited product.
The product is shown in the reaction.
In this reaction leaving group is Cl, the nucleophile is SCH3-
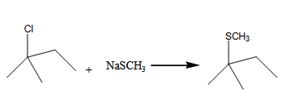
Explanation of Solution
The alkyl halide undergoes E1 reaction with NaOH to yield a
The product is shown here
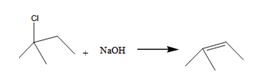
The alkyl halide undergoes SN1reaction with Na+SCH3- to yield a SCH3 Substituited product.
The product is shown in the reaction.
In this reaction leaving group is Cl, the nucleophile is SCH3-

c)
Interpretation:
The expected product between the alkyl halide CH3CH2Cl+Na+SCH3- and NaOH is interpreted.
Answer to Problem 21VC
The benzylchloride undergoes SN1 reaction with Na+SCH3- to yield a product.
The product is shown here.
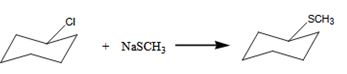
Here the leaving group is Cl.
The nucleophile is SCH3-Hence it undergoes SN1 reaction.
Explanation of Solution
The benzylchloride undergoes E1 reaction with NaOH to yield a product.
The product is shown here
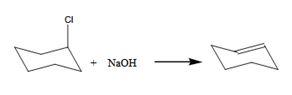
The benzylchloride undergoes SN1 reaction with Na+SCH3- to yield a product.
The product is shown here.
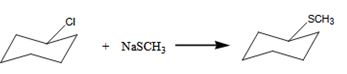
Here the leaving group is Cl.
The nucleophile is SCH3- Hence it undergoes SN1 reaction.
Want to see more full solutions like this?
Chapter 11 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- 2- Give the product for the addition of chlorine when HCl reacts with 3-Hexyne in acetic acid (CH3COOH). Show the correct stereochemistry, (is the product Z or E isomer). Identify the product in the following reaction: CH3CH₂CH₂C=CH + 2Cl₂ 3- What products are obtained by the hydration of the following Alkyne: CH3CH₂CH₂C=CCH₂CH₂CH3 1 ?arrow_forwardPredict the product obtained when 1-pentyne reacts with an excess of HBr:arrow_forwardWrite the appropriate reagents, conditions and products for the following transformations, in a single step. OH II HNO, ? (1) H,SO,arrow_forward
- Provide the structure(s) of the expected major organic product of the reaction shown. 1) Disiamylborane 2) H₂O₂, NaOH OI O II ||| O IV OV CH3CH₂C(CH3)2C=CH OH OH xx xo IVarrow_forwardPredict (by name) the major product for the following reaction. The starting compound is byblo[4.4.0]dec-1-ene. Note the numbering and name the product.arrow_forwardIf phenoxide ion is allowed to react with 1-bromopentane, pentyl phenyl ether is obtained. However, if cyclohexane is used as the alkyl halide, the major products are phenol and cyclohexene. Explain how these products were formed.arrow_forward
- Alkylation of benzene with 1-chlorobutane in the presence of AlCl3 gave not only the expected butylbenzene product but also, as a major product, (1-methylpropyl)benzene. Write an equation for the reaction Propose a mechanism to account for the formation of butylbenzene Propose a mechanism to account for the formation of (1-methylpropyl)benzenearrow_forwardFollowing are two diastereomers of 3-bromo-3,4-dimethylhexane. On treatment with sodium ethoxide in ethanol, each gives 3,4-dimethyl-3-hexene as the major product. One diastereomer gives the E alkene, and the other gives the Z alkene. Which diastereomer gives the Z alkene? Et Me Et Et Et Br Me H Ме b H Br Me aarrow_forwardThe reaction of methylpropene with HBr, under radical conditions, gives two intermediates. Propose a mechanism for the formation of the two products. Propose a mechanism for the following reaction and use electronic factors to account for the formation of a major product: CH2 CH2Br N-Bromosuccinimide (NBS) ho, CCI4 Draw the structure of an antioxidant, Vitamin E free radical and use resonance structures o account for its stability.arrow_forward
- Which functional group(s) would be added to 1-methylcyclohexene using the reagents below: H3O+ hydrogen hydroxyl ketone and aldehyde bromine and hydroxyl bromine and hydrogen bromine hydrogen and hydroxyl aldehyde ketone Save for Laterarrow_forwardWhat products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.arrow_forwardFollowing are diastereomers (A) and (B) of 3-bromo-3,4-dimethylhexane. On treat- ment with sodium ethoxide in ethanol, each gives 3,4-dimethyl-3-hexene as the major product. One diastereomer gives the E alkene, and the other gives the Z alkene. Which diastereomer gives which alkene? Account for the stereoselectivity of each B-elimination. H C-C Mẹ Me Br H Ме Br C-C Me Et Et Et Et (A) (В)arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

