
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
8th Edition
ISBN: 9781305864504
Author: Brent L. Iverson, Sheila Iverson
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.9, Problem DQ
Interpretation Introduction
Interpretation:
The relative amount of trans-tetrol to cis-tetrol at
Concept Introduction:
Ring opening: Ring opening of epoxide takes place by acid-catalyzed hydrolysis of an epoxide.
Acid-catalyzed hydrolysis of an epoxide via
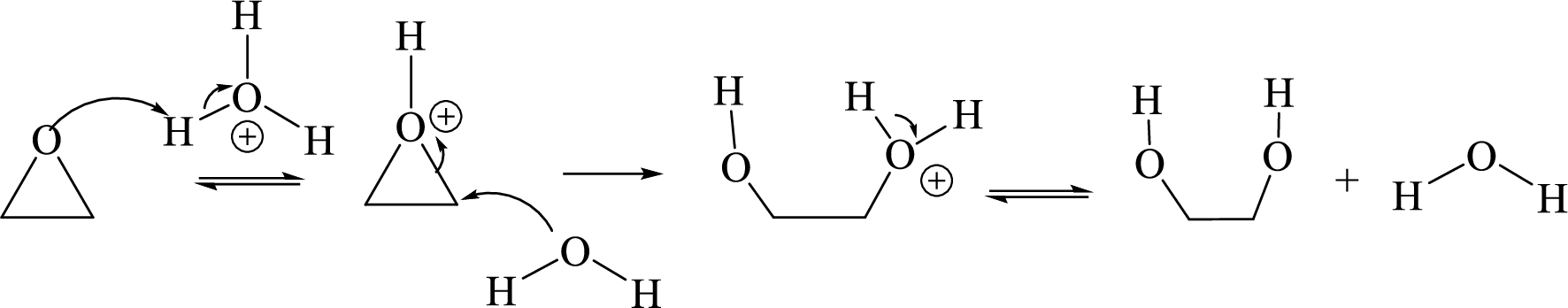
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
7. Complete the following reactions by providing either necessary reagents or products.
CH₂ONa/H+
JUL
NaOH
Br
1). 0₂. 2). (CH₂)₂S
NaOH
CH₂₂/ZnCu
Bry/H₂O
Bry/CC
10. Explain the reaction shown below. Include the following pieces in your explanation.
Identify the electrophile and the nucleophile.
Draw a curved arrow mechanism for this reaction. (Assume an acidic work-up).
Explain the electron flow.
Indicate any regiochemical or stereochemical preferences.
one
b.
c.
d.
LIAIH4
OH
one
5. Propose a synthesis of each of the following compounds using the indicated starting material. You
may use any organic compounds, inorganic compounds, organometallic compounds, or solvents of
your choice. Do not show any reactive intermediates, mechanisms, or transition states, but be sure
to show each isolable compound along your synthetic route.
a.
b.
C.
Ph
+
PPh3 Cl
mylom
Ph.
CH3
steps
steps
steps
H3C.
H3C
Ph
CH3
CH3
Ph
OH
ÕH
Ph
(racemic)
4
Chapter 11 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Ch. 11.2 - Write IUPAC and common names for these ethers. (a)...Ch. 11.3 - Arrange these compounds in order of increasing...Ch. 11.4 - Show how you might use the Williamson ether...Ch. 11.4 - Show how ethyl hexyl ether might be prepared by a...Ch. 11.5 - Account for the fact that treatment of tert-butyl...Ch. 11.5 - Draw structural formulas for the major products of...Ch. 11.6 - Prob. 11.7PCh. 11.8 - Draw the expected products of Sharpless...Ch. 11.9 - Prob. AQCh. 11.9 - Prob. BQ
Ch. 11.9 - Prob. CQCh. 11.9 - Prob. DQCh. 11 - Write names for these compounds. Where possible,...Ch. 11 - Prob. 11.11PCh. 11 - Each compound given in this problem is a common...Ch. 11 - Account for the fact that tetrahydrofuran (THF) is...Ch. 11 - Prob. 11.14PCh. 11 - Write equations to show a combination of reactants...Ch. 11 - Propose a mechanism for this reaction.Ch. 11 - Prob. 11.17PCh. 11 - Prob. 11.18PCh. 11 - Prob. 11.19PCh. 11 - Prob. 11.20PCh. 11 - Ethylene oxide is the starting material for the...Ch. 11 - Prob. 11.22PCh. 11 - Predict the structural formula of the major...Ch. 11 - The following equation shows the reaction of...Ch. 11 - Propose a mechanism to account for this...Ch. 11 - Acid-catalyzed hydrolysis of the following epoxide...Ch. 11 - Prob. 11.27PCh. 11 - Prob. 11.28PCh. 11 - Prob. 11.29PCh. 11 - Propose a mechanism for the following...Ch. 11 - Show reagents and experimental conditions to...Ch. 11 - Starting with cis-3-hexene, show how to prepare...Ch. 11 - Show reagents to convert cycloheptene to each of...Ch. 11 - Show reagents to convert bromocyclopentane to each...Ch. 11 - Prob. 11.35PCh. 11 - Starting with acetylene and ethylene oxide as the...Ch. 11 - Following are the steps in the industrial...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - Aldehydes and ketones react with one molecule of...Ch. 11 - Prob. 11.42PCh. 11 - Write the products of the following sequences of...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - During the synthesis of the antiasthmatic drug...Ch. 11 - Prob. 11.48P
Knowledge Booster
Similar questions
- Draw the structure(s) of the major organic product(s) of the following reaction. H,C. 1. NaOCH3 / CH3OH 2. 1 eq. 2-bromo-2-methylpropane • You do not have to consider stereochemistry. • Omit products derived from the acidic or basic reagent itself, e.g. HN(i-Pr)2 derived from N(i-Pr)2. • If no reaction occurs, draw the organic starting material. • If substantial starting material is present at the end of the reaction, include it in the products. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu.arrow_forwardIn each reaction box, place the best reagent and conditions from the list. NaBH, NINH. -CEN 1) 2) 3) 4) 5) CH,OH. H,O* (cat.) H,O, H₂O, heat 10 (CH,),CHMgBr (excess) acetyl chloride LIAIH acetone H₂O CH, CH,MgBr (excess) acetate ion acetic acid. CH, MgBr (excess)arrow_forward6. Provide reagents and intermediates in the indicated boxes to complete the reaction scheme. + H NaBH3CN, HCI (two steps, no acidic hydrolysis, from one product) 016H (w/o heat) 1. MeLi (xs) 2. HCI, H₂O NH₂ 1. Saponification 2. A 0 1. MeMgBr (xs) 2. HCI, H₂O 018 Me NH₂ 1. Dibal H, 2. H3O+ POCk3, pyr or + 1. LIAIH(O'Bu)3 (XS) 2. acidic workuparrow_forward
- An SN2 mechanism takes place when 3-(bromomethyl)pentane is added to each nucleophile listed below. Which nucleophile will also require an acid-base step after the substitution step in order to produce a neutral (not charged) product? A. LiN(CH3)2 B. NaI C. KN3 D. HSCH3 (Please type answer no write by hend)arrow_forwardWhich condition or sequence is ideal for converting ethyne into the epoxide shown below? A. 1. D2 (1 equivalent)/Pd-C 2. H3CC(O)OOH B. 1. D2/Lindlar 2. H3CC(O)OOH C. 1. H3CC(O)OOH 2. D2/ Pd-C D. 1. Na/ND3 2. H3CC(O)OOHarrow_forwardGg.58.arrow_forward
- C. Draw the structure for the product formed in each of step of the following synthetic sequences. 1. C NH₂ SOCI₂ or P₂O5 or POCI3 (dehydration) 1.CH3MgBr 2.H3O+ 1. NaOH, 12 2. H3O+ + yellow precipitatearrow_forwardPlease don't provide handwriting solutionarrow_forwardChoose the reagents that can be used to perform the following transformation: Select one: a. "OH, H₂O ...OH e. OH b. H3O+ X c. Na₂Cr₂O7, H₂SO4 d. KMnO4, OH Both -OH, H₂O and H3O+ (separately) will work.arrow_forward
- R.arrow_forwardAcyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the tetrahedral intermediate as it is first formed in the following reaction. H3C. ... NH2 HCI/H₂O reflux • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. • Do not include counter-ions, e.g., Na+, I, in your answer. • In cases where there is more than one answer, just draw one. + 4 n [ ?arrow_forwardPp.12. Subject :- Chemistryarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT