
Repeat Prob. 11–56 for a flash chamber pressure of 0.6 MPa.
(a)
The fraction of the refrigerant that evaporates as it is throttled to the flash chamber.
Answer to Problem 57P
The fraction of the refrigerant that evaporates as it is throttled to the flash chamber is
Explanation of Solution
Show the T-s diagram for compression refrigeration cycle as in Figure (1).
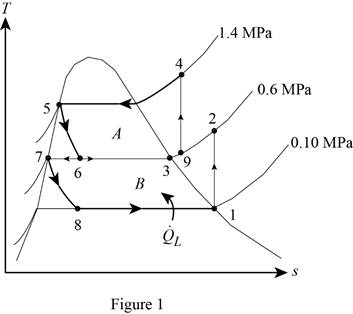
From Figure (1), write the specific enthalpy at state 5 is equal to state 6 due to throttling process.
Here, specific enthalpy at state 5 and 6 is
From Figure (1), write the specific enthalpy at state 7 is equal to state 8 due to throttling process.
Here, specific enthalpy at state 7 and 8 is
Express the fraction of the refrigerant that evaporates as it is throttled to the flash chamber
Here, specific enthalpy at saturated vapor is
Conclusion:
Perform unit conversion of pressure at state 1 from
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the properties corresponding to pressure at state 1
Here, specific entropy and enthalpy at state 1 is
Refer Table A-13, “superheated refrigerant 134a”, and write the specific enthalpy at state 2 corresponding to pressure at state 2 of
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y is specific entropy at state 2 and specific enthalpy at state 2 respectively.
Show the specific enthalpy at state 2 corresponding to specific entropy as in Table (1).
|
Specific entropy at state 2 |
Specific enthalpy at state 2 |
| 0.9500 | 270.83 |
| 0.9519 | |
| 0.9817 | 280.60 |
Substitute
Thus, the specific enthalpy at state 2 is,
Perform unit conversion of pressure at state 3 from
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the property corresponding to pressure at state 3
Perform unit conversion of pressure at state 5 from
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the property corresponding to pressure at state 5
Here, specific enthalpy at saturated liquid is
Substitute
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the property corresponding to pressure at state 8
Substitute
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the specific enthalpy at evaporation and pressure of
Substitute
Hence, the fraction of the refrigerant that evaporates as it is throttled to the flash chamber is
(b)
The rate of heat removed from the refrigerated space.
Answer to Problem 57P
The rate of heat removed from the refrigerated space is
Explanation of Solution
Express the enthalpy at state 9 by using an energy balance on the mixing chamber.
Here, the rate of total energy entering the system is
Express the mass flow rate through the flash chamber.
Here, mass flow rate through condenser is
Express The rate of heat removed from the refrigerated space.
Conclusion:
Substitute
Substitute
Substitute
Hence, the rate of heat removed from the refrigerated space is
(c)
The coefficient of performance.
Answer to Problem 57P
The coefficient of performance is
Explanation of Solution
Express compressor work input per unit mass.
Express the coefficient of performance.
Express entropy at state 4.
Here, specific entropy at state 3 is
Conclusion:
Refer Table A-12, “saturated refrigerant-134a-pressure table”, and write the property corresponding to pressure at state 3
Here, specific entropy at saturated vapor is
Substitute
Refer Table A-13, “superheated refrigerant 134a”, and write the specific enthalpy at state 4 corresponding to pressure at state 4 of
Show the specific enthalpy at state 4 corresponding to specific entropy as in Table (2).
|
Specific entropy at state 4 |
Specific enthalpy at state 4 |
| 0.9389 | 285.47 |
| 0.9444 | |
| 0.9733 | 297.10 |
Use excels and substitute value from Table (2) in Equation (IV) to get,
Substitute
Substitute
Hence, the coefficient of performance is
Want to see more full solutions like this?
Chapter 11 Solutions
THERMODYNAMICS-SI ED. EBOOK >I<
- QIAt the commencement of the compression stroke, the cylinder of an oil engine is charged with air at 96.5kPa and 65°C. Compression takes place to 1/14th of the original volume according to the law PV.35 =C. Fuel the injected, the mass of the fuel injected being 1/40th of that of the air in the cylinder, and combustion takes place at constant pressure. Taking t5he calorific value of the oil as 44000OKJ/kg, determine a- the theoretical pressure and temperature after compression b- the theoretical temperature after combustion c- the fraction of the stroke at which combustion is theoretically complete, cp=1.003KJ/kg.K (3400kPa, 579°C, 1649°C, 0.0967)arrow_forwardA spark ignition cycle having a compression ratio of 9. The air intake is at 100kPa and 20°C, the chamber volume is 500 cm³ before compression stroke and the temperature at the end of adiabatic expansion is 527 °C. Determine (in kPa) the mean effective pressure of the cyclearrow_forwardWhat are the effects of varying the compression ratio on the thermal efficiency and power output of an internal combustion engine?arrow_forward
- What are the design parameters for the various equipment and components (includes the table of specification) used in the refrigeration and cryogenic plant for liquid oxygen production? What are the considerations? What are the design assumptions?arrow_forwardAs a car gets older, will its compression ratio change? How about the mean effective pressure?arrow_forwardThe working substance of a certain Carnot engine is 1.40 mol of an ideal monatomic gas. During the isothermal expansion portion of this engine's cycle, the volume of the gas doubles, while during the adiabatic expansion the volume increases by a factor of 5.7. The work output of the engine is 990 J in each cycle. Part A Compute the temperatures of the two reservoirs between which this engine operates. Express your answer using two significant figures. Enter your answers numerically separated by a comma. IVE| ΑΣΦ TL,TH= Submit Provide Feedback Request Answer ? Karrow_forward
- Calculate the boiler efficiency for a boiler given the following conditions: Composition of solid fuel by mass: C-84%, H=4.5% and O-4.0%, the rest being incombustibles. Composition of air by mass: 0=23% and N=77% Calorific value of fuel: 33.9 MJ/ kg Amount of excess air in combustion: 85 Flue gas temperature: 315.5 °C Ambient temperature: 32.2 °C Specific heat capacity of flue gases: 1.005 kJ/kg K Radiative/convective losses: 2.0% Blowdown and other losses: 2,5%arrow_forward3. Show that the thermal efficiency of the Carnct cycle in terms of the isentropic compression ratio r is given by e = 1-1. k-1arrow_forwardAnswer carefully please. Step by step solution. Determine: (a) the compression ratio. (b) the cutoff ratio. (c) the percent thermal efficiency of the cycle. (d) the mean effective pressure, in kPa.arrow_forward
- What is the steam pressure of ultra-supercritical coal fired plant?arrow_forwardDuring the dry season cooling water is not available for the steam plant in the problem# 1. Consequently the exhaust steam is discharged directly to the atmosphere at 1 bar. Fresh feed water at 30oC is pumped to the boiler. Under these conditions determine the following: a. Dryness fraction of exhaust steam. b. Turbine work output. c. Work input to the feed pump. d. Heat supplied to the boiler. e. Heat rejected to the atmosphere. f. Efficiency of the plant. g. Net power output.arrow_forwardDoes the efficient shaft have a circular cross-section?arrow_forward
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
