
Conceptual Integrated Science
3rd Edition
ISBN: 9780135197394
Author: Hewitt, Paul G., LYONS, Suzanne, (science Teacher), Suchocki, John, Yeh, Jennifer (jennifer Jean)
Publisher: PEARSON EDUCATION (COLLEGE)
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11, Problem 55TE
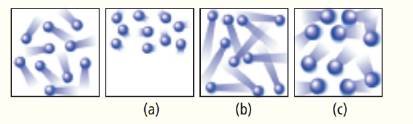
The left most diagram below shows the moving particles of a gas within a rigid container. Which of the three boxes on the right-(a), (b), or (c)-best represents this material upon the addition of heat?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What do you mean by the change of state of matter? Explain:
(a) the change of a solid into a liquid at a constant temperature, and
(b) the change of a liquid into a gas at a constant temperature.
You have a container that can change volumes to keep pressure fixed at 45 lb/in2. The container hold a fixed number of gas molecules (1 mole)
When the temperature of the gas is the container is 20 oC, the volume of the container is 1.7 liters. When the temperature is increased to 45 oC, what is the volume of the container?
Give your answers in liters to one decimal place.
answer true or false for the following
A) In point A, all three phases (solid, liquid, gas) are in equilibrium with each other?
B) Line AB is a solid-liquid equilibrium line?
C) Line AB indicates that the liquid is denser than the solid?
D) Line AC represents the phase changes of sublimation and deposition?
E) Line AD represents the substance of condensing and vaporizing?
pick the right option for the last one
F) State of the substance denoted by point P is?a) solidb) liquidc) vapor
Chapter 11 Solutions
Conceptual Integrated Science
Ch. 11 - Prob. 1RCCCh. 11 - Prob. 2RCCCh. 11 - Prob. 3RCCCh. 11 - Prob. 4RCCCh. 11 - Prob. 5RCCCh. 11 - How are the particles in a solid arranged...Ch. 11 - Which occupies the greatest volume: 1 gram of ice,...Ch. 11 - What is it called when evaporation takes place...Ch. 11 - How is sublimation different from evaporation?Ch. 11 - Prob. 10RCC
Ch. 11 - How much heat is needed to melt 1 gram of ice?...Ch. 11 - What happens to the chemical identity of a...Ch. 11 - What is a physical property? A chemical property?Ch. 11 - What is a chemical bond?Ch. 11 - What changes during a chemical reaction?Ch. 11 - Why is the freezing of water considered to be a...Ch. 11 - Prob. 17RCCCh. 11 - Why is the rusting of iron considered to be a...Ch. 11 - Prob. 19RCCCh. 11 - What is the difference between an element and a...Ch. 11 - How many atoms are in one molecule of H3PO4?Ch. 11 - How many atoms of each element are in one molecule...Ch. 11 - What does the chemical formula of a substance tell...Ch. 11 - Prob. 24RCCCh. 11 - Prob. 25RCCCh. 11 - Prob. 26RCCCh. 11 - What is the chemical formula for the compound...Ch. 11 - Why are common names often used for chemical...Ch. 11 - How soon will nanotechnology give rise to...Ch. 11 - Prob. 30TISCh. 11 - Who is the ultimate expert at nanotechnology?Ch. 11 - Prob. 38TCCh. 11 - Rank these substances in order of increasing...Ch. 11 - Rank the following physical and chemical changes...Ch. 11 - Rank these compounds in order of increasing number...Ch. 11 - How has chemistry influenced our modern...Ch. 11 - While visiting a foreign country, a foreign...Ch. 11 - If someone is able to explain an idea to you using...Ch. 11 - What is the best way to really prove to yourself...Ch. 11 - Prob. 46TECh. 11 - Prob. 47TECh. 11 - What is found between two adjacent molecules of a...Ch. 11 - You combine 50mL of water with 50mL of purified...Ch. 11 - Prob. 50TECh. 11 - Which has stronger attractions among its...Ch. 11 - Prob. 52TECh. 11 - Is it possible for air to be in liquid phase?...Ch. 11 - Prob. 54TECh. 11 - The left most diagram below shows the moving...Ch. 11 - The leftmost diagram here shows two phases of a...Ch. 11 - A cotton ball is dipped in alcohol and wiped...Ch. 11 - A skillet is lined with a thin layer of cooking...Ch. 11 - A cotton ball is dipped in alcohol is wiped across...Ch. 11 - Use exercise 58 as an analogy to describe what...Ch. 11 - Prob. 61TECh. 11 - Prob. 62TECh. 11 - Prob. 63TECh. 11 - Why are physical changes typically easier to...Ch. 11 - Prob. 65TECh. 11 - Prob. 66TECh. 11 - Each night you measure your height just before...Ch. 11 - State whether each of the following is an example...Ch. 11 - State whether each of the following is an example...Ch. 11 - How is sugar dissolving in water an example of a...Ch. 11 - Why is the air over a campfire always moist?Ch. 11 - Prob. 72TECh. 11 - Prob. 73TECh. 11 - Each sphere in the diagrams shown here represents...Ch. 11 - Is aging primarily an example of a physical or a...Ch. 11 - Is nuclear fusion, as described in Chapter 10, an...Ch. 11 - Prob. 77TECh. 11 - Prob. 78TECh. 11 - Oxygen atoms are used to make water molecules....Ch. 11 - Oxygen, O2, is certainly good for you. Does it...Ch. 11 - Prob. 81TECh. 11 - Prob. 82TECh. 11 - Which of the following boxes contains only an...Ch. 11 - Prob. 84TECh. 11 - Prob. 85TECh. 11 - What is the chemical name for a compound with the...Ch. 11 - Prob. 87TECh. 11 - Prob. 88TECh. 11 - Is nanotechnology the result of basic or applied...Ch. 11 - How does a scanning probe microscope differ from...Ch. 11 - People often behave differently in a group...Ch. 11 - Prob. 92TECh. 11 - Medicines, such as pain relievers and...Ch. 11 - Your friend smells cinnamon coming from an...Ch. 11 - The British diplomat, physicist, and...Ch. 11 - Prob. 96TDICh. 11 - A calculator is useful but certainly not exciting....Ch. 11 - How might speculations about potential dangers of...Ch. 11 - Over the past 20 years, the average life...Ch. 11 - Prob. 100TDICh. 11 - Prob. 1RATCh. 11 - The molecules in a small collection of molecules...Ch. 11 - The phase in which atoms and molecules no longer...Ch. 11 - Prob. 4RATCh. 11 - Prob. 5RATCh. 11 - Prob. 6RATCh. 11 - Which is an example of a chemical change? a Water...Ch. 11 - If you burn 50kg of wood and produce 10g of ash,...Ch. 11 - If you have one molecule of TiO2, how many...Ch. 11 - Prob. 10RAT
Additional Science Textbook Solutions
Find more solutions based on key concepts
16.71 CP A turntable 1.50 m in diameter rotates at 75 rpm. Two speakers, each giving off sound of wavelength 31...
University Physics with Modern Physics (14th Edition)
3. What is free-fall, and why does it make you weightless? Briefly describe why astronauts are weightless in th...
The Cosmic Perspective (8th Edition)
8. Light from a sodium lamp (?= 589 nm) illuminates two narrow slits. The fringe spacing on a screen 150 cm beh...
College Physics: A Strategic Approach (4th Edition)
1. Is the universe in space or is space in the universe?
Conceptual Physical Science (6th Edition)
The mass of the suitcase.
Physics (5th Edition)
25. Electric vehicles increase speed by using an electric motor that draws energy from a battery. When the vehi...
College Physics: A Strategic Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- SHOW COMPLETE AND DETAILED SOLUTION. An ideal gas with molecular mass of 24 is at temperature of 90 degrees Celsius and a pressure of 2 bar. What is the density (in kg/m3) of this gas at this condition? A 40 centimeters diamater cylindrical container with a height of 50 centimeters carries a mixture of saturated liquid and saturated vapor water at volume ratio 1:9 liquid to vapor. If the temperature of the system is 100 degrees Celsius, what is the total mass (in kg) of water in the container?arrow_forwardIf you decrease the volume of a gas to 1/3 its original volume, how much bigger or smaller is the pressure of the gas? If you increase the volume to 5 times its original volume, how much bigger or smaller will be the pressure of the gas?arrow_forwardHow do the pressure, temperature, and volume of a gas relate to one another?arrow_forward
- The graph below shows the changes in the temperature of a beaker of ice as it is being heated. Study the graph below and answer the questions that follow. Temperature of water (Boiling) 100°C B →.5 (melting) 15 20 (Freezing) E F 30 34 Time (minutes) Question: How would the volume of ice be different at the end of the 30th minute as compared to its volume at the beginning of the experiment? Why?arrow_forwardNala heated an iron rod for 20 minutes and the length increased from 200 mm to 210 mm. Then he took a thicker iron rod of 200 mm and heated it for 20 minutes also. Would the length of this rod after heating be less than, equal to, or more than 210 mm? Explain your answer.arrow_forward1) Based in your study so far, explain and compare, what happens for a gas like Freon when its compressed to a high pressure ( figure 1) and when its under high tension ( or high expansion ). Discuss what happens to the gas in both situations. (search and use equations and explain) Compressed air and decomprossed air increasing pressure high pressure low volume low pressure high volume O 2012 Encyclopaadia Barrow_forward
- Give descriptions of a solid, a liquid, and a gas in terms of shape and volume.arrow_forwardWould you expect to be larger for a gas or a solid?arrow_forwardThe density or gasoline is 7.30 102 kg/m3 at 0C. Its average coefficient of volume expansion is 9.60 104(C)1 and note that 1.00 gal = 0.003 80 m3. (a) Calculate the mass of 10.0 gal of gas at 0C. (b) If 1.000 m3 of gasoline at 0C is warmed by 20.0C, calculate its new volume. (c) Using the answer to part (b), calculate the density of gasoline at 20.0C. (d) Calculate the mass of 10.0 gal of gas at 20.0C. (e) How many extra kilograms of gasoline would you get if you bought 10.0 gal of gasoline at 0C rather than at 20.0C from a pump that is not temperature compensated?arrow_forward
- what is the density of ideal gas in a room whose volume is 120 cubic meter at 100kpa and 25°carrow_forwardThe combined gas law relates which of the following? A) Volume & Temperature B) Temperature & Pressure C) Volume & Pressure D) Volume, Temperature & Pressurearrow_forwardHow much heat in KJ is required to convert 100 g of solid H2O at - 20 °C into steam at 120 °C? Use the following constants: Trace the diagram. (10 points for diagram and solution) C of water = 4.186 J/g0C C of steam = 1.84 J/g0C C of ice = 2 J/gC0 Lv = 2260J/g LF = 340 J/g *arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY