
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11, Problem 11.46P
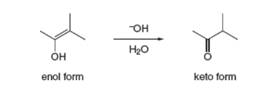
Tautomerization in base resembles tautomerization in acid, but deprotonation precedes protonation in the two-step mechanism. (a) Draw a stepwise mechanism for the following tautomeization. (b) Then draw a stepwise mechanism for the reverse reaction, the conversion of the keto form to the enol.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4. Determine the amount in grams of AgCl (s) formed when 2.580 g AgNO3(s) is added to
45.00 mL of a 0.1250 M CrCl3 (aq) (The other product is aqueous chromium (III) nitrate)
5. Determine the amount (in grams) of Cobalt (II) phosphate formed when an aqueous solution
of 30.0 ml of 0.450 M Sodium Phosphate is mixed with 20.0 mL of 0.500 M aqueous
solution of cobalt (II) nitrate. (The other product is aqueous sodium nitrate)
7. Consider the following reaction that describes the dissolution of copper metal in nitric acid:
Cu (s)
+ 4 HNO3 (aq)
→
Cu(NO3)2 (aq) +
2 H₂O (1)
+
2 NO2 (g)
How many mL of 3.50 M HNO3 (aq) are required to dissolve 20.00 g Cu?
Please correct answer and don't used hand raiting
Chapter 11 Solutions
Organic Chemistry
Ch. 11 - Problem 11.1 Neopheliosyne B is a novel acetylenic...Ch. 11 - Give the IUPAC name for each compound.Ch. 11 - Give the structures corresponding to each of the...Ch. 11 - Prob. 11.4PCh. 11 - Prob. 11.5PCh. 11 - Which bases can deprotonate acetylene? The pKa...Ch. 11 - Draw the organic products formed when each alkyne...Ch. 11 - Draw additional resonance structures for each...Ch. 11 - Problem 11.9 Draw the products formed when is...Ch. 11 - Explain the following result. Although alkenes...
Ch. 11 - Problem 11.11 Draw the keto tautomer of each...Ch. 11 - Prob. 11.12PCh. 11 - a Draw two different enol tautomers of...Ch. 11 - Prob. 11.14PCh. 11 - Problem 11.15 Draw the organic products formed in...Ch. 11 - Problem 11.16 What acetylide anion and alkyl...Ch. 11 - Problem. 11.17 Show how , and can be used to...Ch. 11 - Prob. 11.18PCh. 11 - Draw the products of each reaction. a. b.Ch. 11 - Prob. 11.20PCh. 11 - Problem 11.21 Use retrosynthetic analysis to show...Ch. 11 - Prob. 11.22PCh. 11 - Give the IUPAC name for each compound. a. b.Ch. 11 - Prob. 11.24PCh. 11 - 11.25 Answer the following questions about...Ch. 11 - 11.26 Give the IUPAC name for each alkyne.
a. ...Ch. 11 - Prob. 11.27PCh. 11 - Which of the following pairs of compounds...Ch. 11 - Prob. 11.29PCh. 11 - 11.30 How is each compound related to A? Choose...Ch. 11 - Prob. 11.31PCh. 11 - Prob. 11.32PCh. 11 - 11.33 Draw the products formed when is treated...Ch. 11 - What reagents are needed to convert (CH3CH2)3CCCH...Ch. 11 - Prob. 11.35PCh. 11 - 11.36 What alkynes give each of the following...Ch. 11 - 11.37 What alkyne gives each compound as the only...Ch. 11 - 11.38 Draw the organic products formed in each...Ch. 11 - 11.39 Draw the structure of compounds A-E in the...Ch. 11 - Prob. 11.40PCh. 11 - Prob. 11.41PCh. 11 - 11.42 What reactions are needed to convert alcohol...Ch. 11 - Prob. 11.43PCh. 11 - Prob. 11.44PCh. 11 - 11.45 Explain the following statement. Although ...Ch. 11 - 11.46 Tautomerization in base resembles...Ch. 11 - 11.47 Draw a stepwise mechanism for each...Ch. 11 - Prob. 11.48PCh. 11 - Prob. 11.49PCh. 11 - 11.50 What acetylide anion and alkyl halide are...Ch. 11 - 11.51 Synthesize each compound from acetylene. You...Ch. 11 - 11.52 Devise a synthesis of each compound using ...Ch. 11 - Prob. 11.53PCh. 11 - Prob. 11.54PCh. 11 - 11.55 Devise a synthesis of the ketone, , from ...Ch. 11 - 11.56 Devise a synthesis of each compound using ...Ch. 11 - Prob. 11.57PCh. 11 - Prob. 11.58PCh. 11 - 11.59 N-Chlorosuccinimide (NCS) serves as a source...Ch. 11 - 11.60 Draw a stepwise mechanism for the following...Ch. 11 - 11.61 Draw a stepwise mechanism for the following...Ch. 11 - Prob. 11.62PCh. 11 - 11.63 Write a stepwise mechanism for each of the...Ch. 11 - Prob. 11.64PCh. 11 - 11.65 Explain why an optically active solution of ...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Give the IUPAC name for each compound.
Organic Chemistry
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties (2nd Edition)
2. Which of the following is the best example of the use of a referent? _
a. A red bicycle
b. Big as a dump tru...
Physical Science
Whether two metal foil leaves an electroscope get opposite charge when the electroscope is charged.
Physics of Everyday Phenomena
Why is it unlikely that two neighboring water molecules would be arranged like this?
Campbell Biology (11th Edition)
2. Why is it that the range of resting blood pressures of humans is best represented by a bell-shaped curve co...
Human Biology: Concepts and Current Issues (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used Ai solutionarrow_forward3. An unknown element, X, combines with chlorine to give a substance with the formula XC14. A chlorine analysis of the substance indicates that it contains 83.47% chlorine by mass. What element is X and what is the formula of this compound? (Hint: to identify an element or compound, identify its molar mass. Remember that Molar Mass = (grams A)/(moles A). Solve for each individually and then divide them to find molar mass.)arrow_forward1. When hydrogen sulfide (H2S, MM = 34.08 g/mol) gas is bubbled into a solution of sodium hydroxide (NaOH, 40.00 g/mol), sodium sulfide (Na2S, 78.04 g/mol) and water (18.02 g/mol) are produced according to the balanced chemical equation shown below? H2S 2 NaOH --> Na2S 2 H₂O (a) Assuming the reaction goes to completion, how many grams of sodium sulfide are formed if 2.50g of hydrogen sulfide is bubbled into a solution containing 1.85g of NaOH? (20 pts) (b) Which reactant and how much of it remains after the reaction has been completed? (15 pts) (c) If only 0.400g of sodium sulfide was recovered, what is the percent yield of this reaction (5 pts)arrow_forward
- The organic compound MTBE (methyltertiarybutylether) is used as a fuel additive that allows gasoline to burn more cleanly thus leading to a reduction in pollution. Recently, however, MTBE has been found in the drinking water of a number of communities. As a result several states are phasing out the use of MTBE as a fuel additive. A combustion experiment using 10.00 g of MTBE was found to produce 24.97g of CO2 and 12.26 g of H2O. (a) What is the empirical formula of MTBE assuming it contains C, H, and O only? (b) The molar mass of MTBE was experimentally determined to be 88.1 g/mol. Using this information what is the molecular formula of MTBEarrow_forwardPart 4: Provide a detailed retrosynthetic analysis and a plausible forward synthesis the following molecule. храдо ofarrow_forward3A: Starting with benzocyclobutene, synthesize the naphthalene derivative below.arrow_forward
- 7. The addition of HBr to 2,5-dimethyl-2,4-heptadiene gives the same product, A, at both low and high temperatures. Provide the structure of A and explain the kinetic and thermodynamic product are the same in this reaction. HBr -78°C or 60°C Aarrow_forward3B: Convert the starting material into the chiral epoxytriol below. OH OH = OH OHarrow_forward3D: Convert the aromatic triketone to the 1,3,5-triethylcyclohexane shown below. ہوئےarrow_forward
- Indicate how to find the energy difference between two levels in cm-1, knowing that its value is 2.5x10-25 joules.arrow_forwardThe gyromagnetic ratio (gamma) for 1H is 2.675x108 s-1 T-1. If the applied field is 1,409 T what will be the separation between nuclear energy levels?arrow_forwardChances Ad ~stract one 11. (10pts total) Consider the radical chlorination of 1,3-diethylcyclohexane depicted below. 4 • 6H total $4th total Statistical pro 21 total 2 H A 2H 래 • 4H totul < 3°C-H werkest bund - abstraction he leads to then mo fac a) (6pts) How many unique mono-chlorinated products can be formed and what are the structures for the thermodynamically and statistically favored products? рос 6 -વા J Number of Unique Mono-Chlorinated Products Thermodynamically Favored Product Statistically Favored Product b) (4pts) Draw the arrow pushing mechanism for the FIRST propagation step (p-1) for the formation of the thermodynamically favored product. Only draw the p-1 step. You do not need to include lone pairs of electrons. No enthalpy calculation necessary H H-Clarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY