
Concept explainers
(a)
Interpretation:
The increasing order of acidity of the alcohols is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the alcohols is given below.
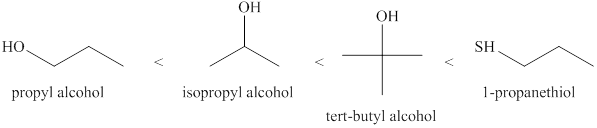
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing hydrogen ion.
Sulfur atom present in
Order of the acidity is shown below in Figure 1.

Figure 1
The increasing order of acidity of the molecules is
(b)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is given below.
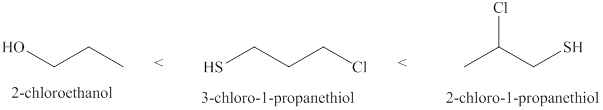
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing hydrogen ion. Sulfur atom present in
Therefore, increasing order of the given molecules is shown below in Figure 2.
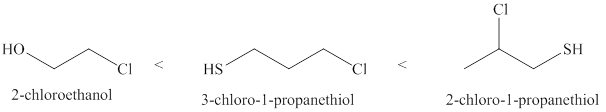
Figure 2
The increasing order of acidity of the given molecules is
(c)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is stated below.
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing hydrogen ion. Nitrogen atom which is positively charged, stabilizes the negative charge which is generated after releasing the hydrogen ion. Therefore, structure 3 is the most stable structure with most acidic character. Position of the electronegative atom also determines the strength of an acid. Closer the electronegative atom to the generated negative charge after releasing the hydrogen ion, more is the strength of the acid.
Therefore, increasing order of the given molecules is stated below.
The increasing order of acidity of the given molecules is
(d)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is stated below.
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing the hydrogen ion. In structure
Therefore, increasing order of acidity of the given molecules is stated below.
The increasing order of acidity of the given molecules is
(e)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is stated below.
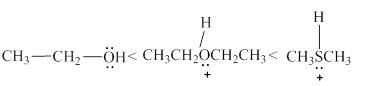
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing the hydrogen ion. In structure
Therefore, increasing order of the given molecules is stated below in Figure 3.
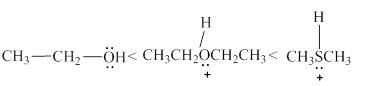
Figure 3
The increasing order of acidity of the given molecules is shown above in Figure 3.
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry
- Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC≡CH, (CF3)2CHOH, CH3OH (b) Phenol, p-methylphenol, p-[trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxybenzonic acidarrow_forwardExplain this statement: Although 2-methoxyacetic acid (CH3OCH2COOH) is a stronger acid than acetic acid (CH3COOH), p-methoxybenzoic acid (CH3OC6H4COOH) is a weaker acid than benzoic acid (C6H5COOH).arrow_forwardExplain this statement: Although 2-methoxyacetic acid (CH3OCH2COOH)is a stronger acid than acetic acid (CH3COOH), p-methoxybenzoic acid(CH3OC6H4COOH) is a weaker acid than benzoic acid (C6H5COOH).arrow_forward
- The following pKa values have been measured. Explain why a hydroxyl group in the para position decreases the acidity while a hydroxyl group in the meta position increases the acidity.arrow_forward18-28 Arrange these compounds in order of increasing acidity: benzoic acid, benzyl alcohol, phenol.arrow_forwardArrange these compounds in order of increasing acidity: 2,4-dichlorophenol, phenol, cyclohexanol.arrow_forward
- Will acetylene react with sodium hydride according to the following equation to form a salt and hydrogen, H2? Using pKa values given in Table 4.1, calculate Keq for this equilibrium.arrow_forward1. Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do. HF NH3 H2SO4 CH3OH CH3COOH H3O+ H2O2. Consider the following compounds that vary from nearly nonacidic to strongly acidic. Draw the conjugate bases of these compounds, and explain why the acidity increases so dramatically with substitution by nitro groups. CH4 CH3NO2 CH2(NO2)2 CH(NO2)3arrow_forwardArrange these compounds in order of increasing acidity: 2,4-dichlorophenol, phenol, cyclohexanol.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



