
To find:
a) Describe the changes in
b) Explain why pressure does not change during the
c) Explain why the pressure is decreased during
d) Explain why the pressure is increased during the
e) Explain if the root-mean-square speed of the molecule in the
f) Explain why the molecules in
Answer to Problem 10.20VP
Solution:
a)
In transition
In transition
In transition 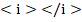 increases.
increases.
In transition
In transition
In transition
In transition
In transition
b) If
c) In transitions
d) Temperature is increasing. Therefore, it will increase the pressure for
e) In
f) More molecules are moving at the root-mean-square speed in [E].
Explanation of Solution
a) As per
In transition
In transition
In transition
In transition
In transition
In transition
In transition
In transition
b) According to Charles’s law,
So, if
c) According to the Boyle’s law,
d) According to the Amonton’s law, temperature is increasing. Therefore, it will increase the pressure for
e) In
f) The fraction of molecules moving at the root-mean-square speed increases as the temperature decreases. So, more molecules are moving at the root-mean-square speed in [E].
Conclusion:
We use the changes in each given transition and different gas laws to find out the changes in pressure, volume, moles, and temperature in all transitions.
Want to see more full solutions like this?
Chapter 10 Solutions
Chemistry: An Atoms-Focused Approach (Second Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





