
Concept explainers
(a)
Interpretation:
The empirical formula of the given hydrocarbon has to be determined.
Concept Introduction:
The hydrocarbon compounds are compounds which contains only carbon and hydrogen atoms. The combustion of hydrocarbon compounds produces large amount of heat. Combustion reaction of a hydrocarbon results in the formation of carbon dioxide and water.
(a)
Answer to Problem 101QRT
The empirical formula of given hydrocarbon is
Explanation of Solution
The mass of carbon dioxide produced is
The mass of water produced is
The molar mass of carbon dioxide is
The molar mass of water is
Use the expression to calculate number of moles.
Substitute
Therefore, the number of moles of carbon dioxide is
Consider the combustion reaction of hydrocarbon as follows.
Here,
When combustion of hydrocarbon takes place, the carbon atoms present in hydrocarbon gets converted to carbon dioxide molecule. One molecule of carbon dioxide contains one carbon atom. Therefore, the number of moles of carbon atoms present in hydrocarbon is equal to the moles of carbon dioxide.
Therefore, moles of carbon atom in hydrocarbon is
Substitute
Therefore, number of moles of water is
When the combustion of hydrocarbon takes place the hydrogen atoms present in hydrocarbon gets converted into hydrogen atoms of water molecule. One molecule of water contains two hydrogen atoms. Therefore, the number of moles of hydrogen atom present in hydrocarbon is twice the number of moles of water.
Therefore, the number of moles of hydrogen atoms is
The smallest number of moles is
Use the expression to calculate mole ratio.
Substitute
Substitute
Therefore, the empirical formula of given hydrocarbon is
(b)
Interpretation:
The given hydrocarbon is an
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 101QRT
The given hydrocarbon compound is an alkene.
Explanation of Solution
The general formula of alkane is
(c)
Interpretation:
The Lewis structure for given hydrocarbon has to be drawn.
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 101QRT
The Lewis structure for given hydrocarbon is as follows.
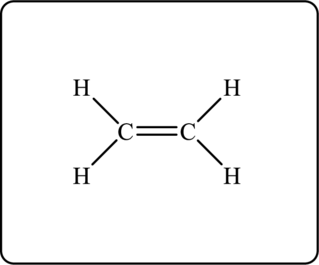
Another possible structure is as follows.
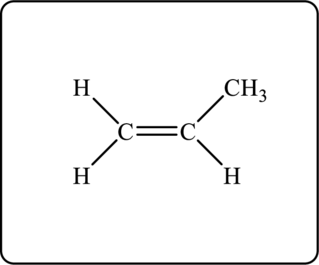
Explanation of Solution
The given hydrocarbon has empirical formula
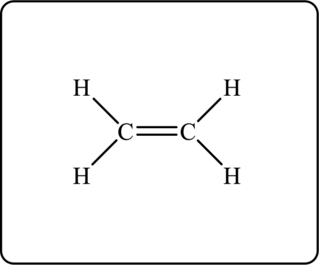
Figure 1
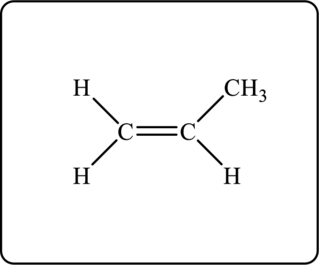
Figure 2
Want to see more full solutions like this?
Chapter 10 Solutions
Bundle: Chemistry: The Molecular Science, 5th, Loose-Leaf + OWLv2 with Quick Prep 24-Months Printed Access Card
- Complete and balance the following combustion reactions. Assume that each hydrocarbon is converted completely to carbon dioxide and water. (a) Propane + O2 (b) Octane + O2 (c) Cyclohexane + O2 (d) 2-Methylpentane + O2arrow_forward(a) What is the difference between chlorofluorocarbons and hydrofluorocarbons?arrow_forwardPropane, C3H8, is a hydrocarbon that is commonly used as a fuel.(a) Write a balanced equation for the complete combustion of propane gas.(b) Calculate the volume of air at 25 °C and 1.00 atmosphere that is needed to completely combust 25.0 grams of propane. Assume that air is 21.0 percent O2 by volume. (Hint:use the information that 1.00 L of air at 25 °C and 1.00 atm contains 0.275 g of O2 per liter.)(c) The heat of combustion of propane is −2,219.2 kJ/mol. Calculate the heat of formation, ΔH°f of propane given that ΔH°f of H2O(l) = −285.8 kJ/mol and ΔH°f of CO2(g) = −393.5 kJ/mol. (d) Assuming that all of the heat released in burning 25.0 grams of propane is transferred to 4.00 kilograms of water, calculate the increase in temperature of the water.arrow_forward
- Alcohols A, B, and C all have the composition C4H10O. Molecules of alcohol A contain a branched carbon chain and can be oxidized to an aldehyde; molecules of alcohol B contain a linear carbon chain and can be oxidizedto a ketone; and molecules of alcohol C can be oxidized to neither an aldehyde nor a ketone. Write the Lewis structures of these molecules.arrow_forwardWhat structural features help us identify a compound as(a) an alkane, (b) a cycloalkane, (c) an alkene, (d) an alkyne,(e) a saturated hydrocarbon, (f) an aromatic hydrocarbon?arrow_forward(a) What structural feature is associated with each type of hydrocarbon: alkane, cycloalkane, alkene, and alkyne?(b) Give the general formula for each type.(c) Which hydrocarbons are considered saturated?arrow_forward
- (a) What is meant by the term isomer ? (b) Among the four alkanes, ethane, propane, butane, and pentane, which is capable of existing in isomeric forms?arrow_forwardThis question is about the chemistry of alkenes, which are unsaturated hydrocarbons. (a) State what is meant by the term unsaturated as applied to a hydrocarbon. (1) (b) An organic compound, X, is an unsaturated hydrocarbon with molecular formula CH₂. (i) Draw the displayed formulae and give the names of two molecules with molecular formula C₂H, which are E/Z isomers. (3) Isomer 1 Isomer 2 Name: Name:arrow_forwardOctane 1C8H182 is a liquid hydrocarbon at room temperature that is a constituent of gasoline. (a) Write a balanced equation for the combustion of C8H181l2 to form CO21g2 and H2O1l2.arrow_forward
- (a) (b) Define the terms 'functional group' and 'unsaturated hydrocarbon'. Name the following organic compounds: i. ii. (c) Draw skeleton structures for the following organic compounds: i. 3,4-dimethylheptane 3-methylheptanal ii. OH (d) The following two organic compounds are structural isomers to each other. Carefully identify and justify the structural isomers type (skeletal, functional, or positional) with their common molecular formula. HO O HO nos. A Barrow_forwardTRUE OR FALSE (a) A functional group is a group of atoms in an organic molecule that undergoes a predictable set of chemical reactions. (b) The functional group of an alcohol, an aldehyde, and a ketone have in common the fact that each contains a single oxygen atom. (c) A primary alcohol has one —OH group, a secondary alcohol has two —OH groups, and a tertiary alcohol has three —OH groups. (d) There are two alcohols with the molecular formula C3H8O. (e) There are three amines with the molecular formula C3H9N. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic…arrow_forwardWrite a balanced chemical reaction for the incomplete combustion of pentane, C5H12C5H12 in oxygen gas to form carbon monoxide gas as one of the products.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning


