
(a)
Interpretation:
The mechanism for the given reaction is to be drawn.
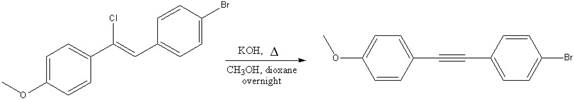
Concept introduction:
The substrates having the leaving group in a vinylic position are quire resistant to nucleophilic substitution and elimination reactions. The four electrons of the
(b)
Interpretation:
The mechanism for the given reactions is to be drawn.
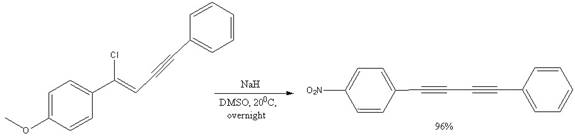
Concept introduction:
The substrates having the leaving group in a vinylic position are quire resistant to nucleophilic substitution and elimination reactions. The four electrons of the
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- How might nucleophilic catalysis work?Draw out a possible mechanism.arrow_forwardYour answer is incorrect. Predict the relative rates of these reactions. That is, select 1 next to the reaction with the fastest rate, 2 next to the reaction with the next fastest rate, and so on. Note for advanced students: you may assume these reactions all take place in a polar aprotic solvent, like DMSO. Incorrect Your answer is incorrect. Predict the relative rates of these reactions. That is, select I next to the reaction with the fastest rate, 2 next to the reaction with the next fastest rate, and so on. Note for advanced students: you may assume these reactions all take place in a polar aprotic solvent, like DMSO. Reaction Relative Rate G + H₂O - OH₂ 2 Br + H₂S - + Br 3 + H₂S SHI + ci I (fastest) Br + H₂O OM₂ + Br 4 (slowest)arrow_forwardThe following reaction is believed to proceed through the intermediate shown. Draw the complete, detailed mechanism that leads to the formation of that intermediate. NH, NH3 HO НО НО OH `NH2arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
