
Concept explainers
Interpretation:
The equation that shows the reaction of the given acid with water by considering the Bronsted-Lowry acid-base theory is to be written. All the electron pairs, formal chargers and curved arrows that represent the electron movement in the respective reaction are to be shown.
Concept introduction:
An acid is a chemical substance that readily donates protons and a base is a chemical substance that can easily accept a proton. During an acid-base reaction, the interaction between an acid and a base is taken place because of the transfer of a proton. The stronger the acid, the smaller its
Answer to Problem 63P
Solution:
a)
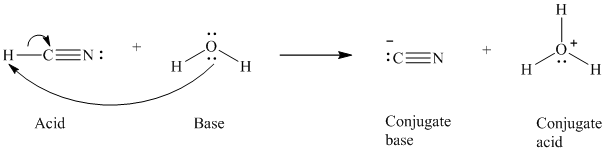
The formal charge on the the oxygen atom is
b)
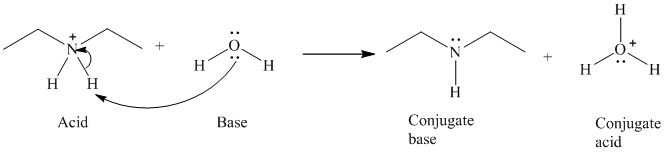
The formal charge on both the the oxygen atom and the nitrogen atom is
c)
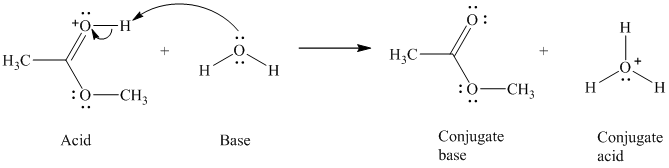 The formal charge on the oxygen atom is
The formal charge on the oxygen atom is
Explanation of Solution
a) The reaction of an acid with water.
In the respective reaction, the water acts as a base. The unshared electron pair of oxygen atom present in water is used to remove the proton from the acid. Water, after accepting the proton, is converted to its conjugate acid, that is, hydronium ion and the acid is converted to its conjugate base.
The curved arrows showing the electron movement is given below:
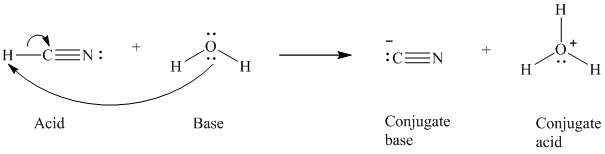
The formula that is used to calculate the electron count on the conjugate base is as follows:
Substitute
The formula that is used to calculate the formal charge on the oxygen atom is as follows:
Substitute
b) The reaction of an acid with water.
The given acid reacts with water. So water acts as a base. The unshared electron pair of the oxygen atom in water is used to remove the proton from the acid. Water, after accepting the proton is converted to its conjugate acid, that is, hydronium ion and the acid is converted to its conjugate base.
The curved arrows showing the electron movement are shown below:
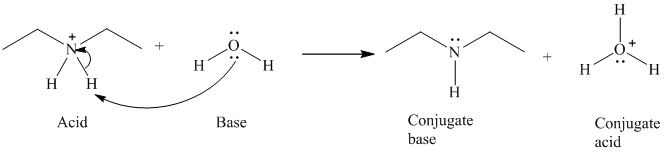
The formula that is used to calculate the electron count on the nitrogen atom is as follows:
Substitute
The formula that is used to calculate the formal charge on the nitrogen atom is as follows:
Substitute
For calculating formal charge on the oxygen atom, recall the electron count formula:
Substitute
Recall the formula for formal charge:
Substitute
c) The given acid reacts with water.
So water acts as a base. The unshared electron pair of the oxygen atom in water is used to remove the proton from the acid. Water, after accepting the proton, is converted to its conjugate acid, that is, hydronium ion and the acid is converted to its conjugate base.
The curved arrows showing the electron movement are shown below:
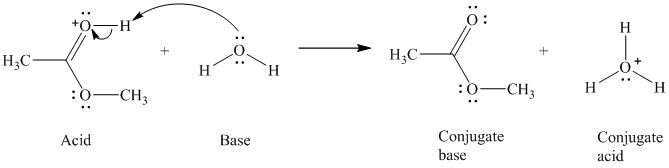
The formula that is used to calculate the electron count on the oxygen is as follows:
Substitute
The formula that is used to calculate the formal charge on the oxygen atom is as follows:
Substitute
Want to see more full solutions like this?
Chapter 1 Solutions
ORGANIC CHEMISTRY (LOOSELEAF)-PACKAGE
- Unshared, or lone, electron pairs play an important role in determining the chemical and physical properties of organic compounds. Thus, it is important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. The number of unshared pairs at atom a is © The number of unshared pairs at atom b is The number of unshared pairs at atom c is 0. 0 e The number of unshared pairs at atom a is The number of unshared pairs at atom b is The number of unshared pairs at atom c is e.arrow_forwardWhich atom bears the formal positive charge in the hydronium ion?arrow_forwardDraw the Lewis structure created by the curved arrow. Write in formal charges wherever they are not equal to zero. H C=arrow_forward
- Complete the following acid-base reaction, using curved arrows to show the movement of electrons. Write Lewis structures for all reactants and products. Label all acids and bases. Predict the position of the equilibrium and explain your reasoning. NH ноarrow_forwardQUESTION 4. (a) Below are two structural isomers. Interestingly, one of these structures is significantly more acidic than the other. Circle the most acidic proton in each. Determine which structure is more acidic and explain why. Use your knowledge of resonance structures and molecular geometry to support your argument. NO₂ NO₂ OH OHarrow_forwardYou will not find “hydroxide” in the stockroom, but you will find sodium hydroxide (NaOH) andpotassium hydroxide (KOH). Lithium hydroxide (LiOH) is expensive and used in spacecraft airfilters since hydroxide reacts with carbon dioxide, and lithium is lighter than sodium or potassium.Cesium and francium hydroxides are very expensive and little used. Is this information consistentwith your answer to the previous question?arrow_forward
- Ethanol, , is a compound in which the formal charge on all the atoms is zero. Under certain conditions the bond can be broken so that both electrons remain with the oxygen atom. The products are In this structure the oxygen owns one electron from shared pair and two electrons from each of unshared pairs. The total number of electrons belonging to oxygen is Oxygen is a Group element. The formal charge on the oxygen atom is . The correct Lewis structure for the ethoxide ion is Note that the other fragment, the proton, leaves with a formal charge of +1.arrow_forwardThe carbon atom owns one electron from each of shared pairs and two electrons from unshared pair. The number of electrons that belong to carbon is . Carbon is a Group element. Since the carbon atom has one more electron than it would in the neutral, unbonded state, it has a formal charge of –1 . The Lewis structure for the methyl anion is The lithium fragment must have a formal charge ofarrow_forwardThe oxygen atom in acetone possesses ____ unshared pairs and ____ shared pairs of electrons. The number of electrons that belong to oxygen is ____. Oxygen is a Group ____ element. The formal charge on oxygen in acetone is ____.arrow_forward
- Unshared, or lone, electron pairs play an important role in determining the chermical and physical properties of organic compounds. Thus, it is important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. The number of unshared pairs at atom a is The number of unshared pairs at atom b is HyC CH The number of unshared pairs at atom e is The number of unshared pairs at atom a is The number of unshared pairs at atom b is HC CH The number of unshared pairs at atom e isarrow_forwardMany free radicals combine to form molecules that do not contain any unpaired electrons. The driving force for the radical-radical combination reaction is the formation of a new electron-pair bond. Consider the formation of hydrogen peroxide. 2 OH(g) - H,O,(g) Write Lewis formulas for the reactant and product species in the chemical equation. Include nonbonding electrons. OH(g) H,O,(g) H H. : 0 H Question Source: MRG - General Chem about us privacy policy terms of use help contact us careers : :arrow_forwardCan someone please show me how the all the transfer of electrons occurs using arrows and writing out the formal charges. I'm particularly confused about the first line. Please show every single electron pair involved (even on the h2o).arrow_forward

 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



