
Interpretation:
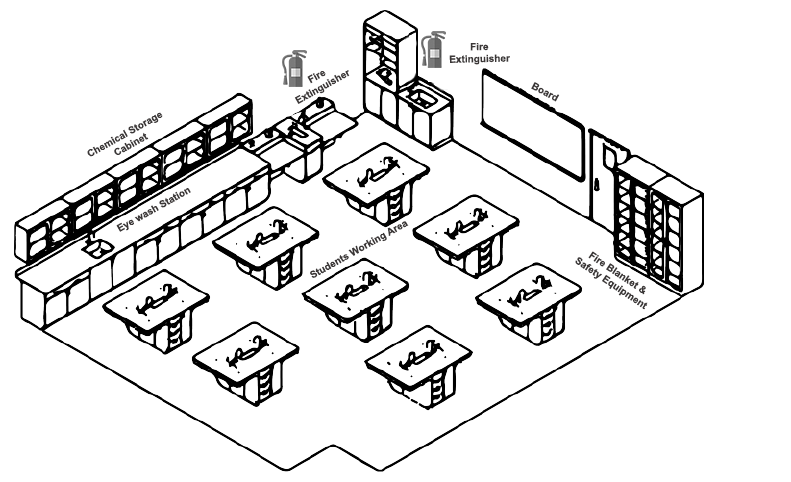
Rough sketch of the lab is to be drawn and also mark where the fire extinguishers, fire blanket, eye wash station, and other safety equipment are, as well as where you’ll be working.
Concept Introduction:
A chemistry laboratory is a facility that provides controlled conditions in which chemistry experiments, research and measurement may be performed.
A chemist might use a wet laboratory and it contains various inherent toxic and hazardous chemicals like acids, alkali compounds, etc.
Answer to Problem 1E
The fire extinguishers, fire blanket, eye wash station, and other safety equipments are shown in the rough sketch of lab.
Explanation of Solution
A lab instructor must give details of experiments to the students and give them lab report format so they will accomplish according to procedure. When students are performing experiment, instructor should tell them to write all observation and results with good explanations. It provides you proper knowledge and better understanding about the experiment, chemical and handling of the chemicals. As a chemistry student, one should follow the safety instruction which is given by instructor.
Rough sketch of the lab is drawn below:
Want to see more full solutions like this?
Chapter 1 Solutions
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
- Using the lab instructions and data table with data, please help me complete part 1 and 2 of my lab worksheetarrow_forwardI completed my pre-lab and want it reviewed for correctionsarrow_forwardIN THE EVENT OF A FIRE, SEVERE WEATHER OR ANY OTHER EVENT REQUIRING LAB EVACUATION.... A. STABALIZE REACTIONS IN PROGRESS.B. CLOSE FUME HOOD SASHES.C. CLOSE THE LAB DOOR.D. ALL OF THE ABOVE.arrow_forward
- Essay about correct use and handling of lab equipment?arrow_forwardwhich is NOT appropriate for lab and why not? chemical resistant goggles, eyeglasses, or safety glassesarrow_forwardWhat are the uses of Laboratory Apparatus below: 1. Aspirator or Pipette Bulb 2. Evaporating Dish 3. Mortar and Pestle 4. Graduated Pipette 5. Acid Burette 6. Spatula 7. Stirring Rodarrow_forward
- 1.Explain the slogan about chemical safety " A spill, A slip, A hospital trip" 2.How do you apply 5s(sort, set in order, shine, standardize, sustain) to attain chemical safety at home and in school?. Enumeratearrow_forwardTo ensure that you are not confused, I attached what the experiment was about. I highly recommend you read through it before going to the questions. file:///Users/michaelzheng/Downloads/ExploringEnergy%20(2).pdf Only answer Post lab questions 2-5 For number five, put the answers in the format stated in the background. Thank you.arrow_forwardCarbon monoxide levels and the presence of soot can beused to determine whether the individual was___________ at the time of the fire.arrow_forward
- Suzy Baker What three areas are affected by an "irritant"? (refer to ACS Safety in the Academic Chemistry Laboratories Vol. 1) [use the answer format= ans 1, ans 2, ans 3]arrow_forwardread the Procedure and answer the post lab questions below: Using a balance, mass between 1.50 – 2.00 grams of sodium carbonate in a pre-massed 150mL beaker. Add 20 mL of distilled water and stir thoroughly to make sure all the crystals are dissolved. Rinse the stirring rod with a little distilled water after stirring. Using a balance, mass between 1.50 – 2.00 grams of calcium chloride dihydrate in a pre-massed 50 mL beaker. Repeat Step 2 for the solution in the 50 mL beaker. Pour the calcium chloride solution into the 150mL beaker containing the sodium carbonate solution and stir. Mass a piece of filter paper. Fold the filter paper and place it into the funnel. Wet it with a little distilled water to ensure that it is stuck to the sides of the funnel. Slowly pour the mixture from the beaker into the funnel and collect the filtrate into a 125mL erlenmeyer flask. Do not allow the mixture to rise above the edge of the filter paper. Rinse the beaker with about 10 mL of distilled water.…arrow_forwardread the Procedure and answer the post lab questions below: Using a balance, mass between 1.50 – 2.00 grams of sodium carbonate in a pre-massed 150mL beaker. Add 20 mL of distilled water and stir thoroughly to make sure all the crystals are dissolved. Rinse the stirring rod with a little distilled water after stirring. Using a balance, mass between 1.50 – 2.00 grams of calcium chloride dihydrate in a pre-massed 50 mL beaker. Repeat Step 2 for the solution in the 50 mL beaker. Pour the calcium chloride solution into the 150mL beaker containing the sodium carbonate solution and stir. Mass a piece of filter paper. Fold the filter paper and place it into the funnel. Wet it with a little distilled water to ensure that it is stuck to the sides of the funnel. Slowly pour the mixture from the beaker into the funnel and collect the filtrate into a 125mL erlenmeyer flask. Do not allow the mixture to rise above the edge of the filter paper. Rinse the beaker with about 10 mL of distilled water.…arrow_forward
 World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
