
Concept explainers
Write Lewis structures for these ions. Show all valence electrons and all formal charges.
- (a) Amide ion, NH2−
- (b) Bicarbonate ion, HCO3−
- (c) Carbonate ion, CO32−
- (d) Nitrate ion, NO3−
- (e) Formate ion, HCOO−
- (f) Acetate ion, CH3COO−
(a)
Interpretation:
Lewis structure of the ions, valence electron and formal charge has to be identified.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Valence shell:
The outer most shell of the element is called as valence shell.
Valence electron:
The electrons present in the outer shell are called valence electron.
For example:
Nitrogen element has seven electrons and it has two shells, the second shell is the valence shell and it has five electrons in the valence shell. The five electrons are called as valence electrons.
Formal charge:
The charge on an atom in a molecule is called its formal charge.
Formal charge can be calculated by using following formula,
Explanation of Solution
The given ion is amide ion,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined. The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it. Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
The valence electron for the hydrogen atom is one, the valence electron for the nitrogen is five, therefore, and the Lewis structure of the ion is given below,
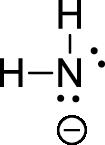
Formal charge can be calculated by using following formula,
(b)
Interpretation:
The Lewis structure of the ions, valence electron and formal charge has to be identified.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Valence shell:
The outer most shell of the element is called as valence shell.
Valence electron:
The electrons present in the outer shell are called valence electron.
For example:
Nitrogen element has seven electrons and it has two shells, the second shell is the valence shell and it has five electrons in the valence shell. The five electrons are called as valence electrons.
Formal charge:
The charge on an atom in a molecule is called its formal charge.
Formal charge can be calculated by using following formula,
Explanation of Solution
The given ion is bicarbonate ion,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined. The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it. Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
The valence electron for the hydrogen atom is one, the valence electron for the carbon is four and valence electron of oxygen is six, therefore, and the Lewis structure of the ion is given below,
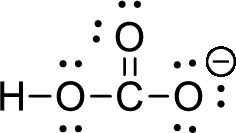
Formal charge can be calculated by using following formula,
(c)
Interpretation:
The Lewis structure of the ions, valence electron and formal charge has to be identified.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Valence shell:
The outer most shell of the element is called as valence shell.
Valence electron:
The electrons present in the outer shell are called valence electron.
For example:
Nitrogen element has seven electrons and it has two shells, the second shell is the valence shell and it has five electrons in the valence shell. The five electrons are called as valence electrons.
Formal charge:
The charge on an atom in a molecule is called its formal charge.
Formal charge can be calculated by using following formula,
Explanation of Solution
The given ion is carbonate ion,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined. The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it. Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
The valence electron the carbon is four and valence electron of oxygen is six, therefore, and the Lewis structure of the ion is given below,
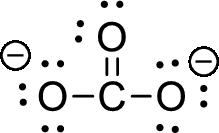
Formal charge can be calculated by using following formula,
(d)
Interpretation:
The Lewis structure of the ions, valence electron and formal charge has to be identified.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Valence shell:
The outer most shell of the element is called as valence shell.
Valence electron:
The electrons present in the outer shell are called valence electron.
For example:
Nitrogen element has seven electrons and it has two shells, the second shell is the valence shell and it has five electrons in the valence shell. The five electrons are called as valence electrons.
Formal charge:
The charge on an atom in a molecule is called its formal charge.
Formal charge can be calculated by using following formula,
Explanation of Solution
The given ion is nitrate ion,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined. The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it. Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
The valence electron for the nitrogen atom is five, the valence electron for the carbon is four and valence electron of oxygen is six, therefore, and the Lewis structure of the ion is given below,
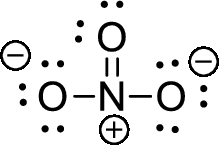
Formal charge for nitrogen atom can be calculated by using following formula,
Formal charge for oxygen can be calculated by using following formula,
(e)
Interpretation:
The Lewis structure of the ions, valence electron and formal charge has to be identified.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Valence shell:
The outer most shell of the element is called as valence shell.
Valence electron:
The electrons present in the outer shell are called valence electron.
For example:
Nitrogen element has seven electrons and it has two shells, the second shell is the valence shell and it has five electrons in the valence shell. The five electrons are called as valence electrons.
Formal charge:
The charge on an atom in a molecule is called its formal charge.
Formal charge can be calculated by using following formula,
Explanation of Solution
The given ion is formate ion,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined. The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it. Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
The valence electron for the hydrogen atom is one, the valence electron for the carbon is four and valence electron of oxygen is six, therefore, and the Lewis structure of the ion is given below,
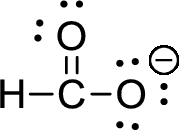
Formal charge can be calculated by using following formula,
(f)
Interpretation:
The Lewis structure of the ions, valence electron and formal charge has to be identified.
Concept introduction:
Lewis structure: The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell. Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance. All the possible resonance structures are imaginary whereas the resonance hybrid is real. These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Valence shell:
The outer most shell of the element is called as valence shell.
Valence electron:
The electrons present in the outer shell are called valence electron.
For example:
Nitrogen element has seven electrons and it has two shells, the second shell is the valence shell and it has five electrons in the valence shell. The five electrons are called as valence electrons.
Formal charge:
The charge on an atom in a molecule is called its formal charge.
Formal charge can be calculated by using following formula,
Explanation of Solution
The given ion is acetate ion,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined. The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it. Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
The valence electron for the hydrogen atom is one, the valence electron for the carbon is four and valence electron of oxygen is six, therefore, and the Lewis structure of the ion is given below,
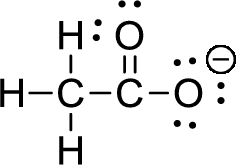
Formal charge can be calculated by using following formula,
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry
- Consider the formate ion, HCO2", which is the anion formed when formic acid loses an H* ion. The H and the two O atoms are bonded to the central C atom. (a) Draw the best Lewis structure(s) for this ion. (b) Are resonance structures needed to describe the structure? Explain briefly (c) Would you predict that the C-O bond lengths in the formate ion would be longer or shorter relative to those in CO2? Explain brieflyarrow_forwardWrite Lewis dot symbols for the following atoms and ions. (a) I (b) I− (c) S (d) S2−arrow_forwardIn each case, tell whether the bond is ionic, polar cova- lent, or nonpolar covalent. (a) Br, (e) SiH, (d) SrF, (c) HCl (g) N, (b) BrCl 2 (f) CO (h) CsCl 4.arrow_forward
- Cyanogen (CN)2 is known as pseodohalogen because it has some properties like halogens. It is composed of two CN’s joined together.(i) Draw the Lewis structure for all the possible combination for (CN)2.(ii) Calculate the formal charge and determine which one of the structures that you have drawn is most stable.(iii) For the stable structure, determine the geometry around the two central atoms.(iv) For the stable structure, draw the dipole arrows for the bonds.(v) Base on the stable structure, determine the polarity of molecule and state your reason.arrow_forwardWrite Lewis structures for the following molecules and ions: (d) CH3COO−, (e) CN−, (f) CH3CH2NH3+.arrow_forwardA stable triatomic molecule can be formed that contains one atom each of nitrogen, sulfur, and fluorine. Three bonding structures are possible, depending on which is the central atom: NSF, SNF, and SFN. (a) Write a Lewis diagram for each of these molecules, indicating the formal charge on each atom. (b) Often, the structure with the least separation of formal charge is the most stable. Is this statement consistent with the observed structure for this molecule— namely, NSF, which has a central sulfur atom? (c) Does consideration of the electronegativities of N, S, and F from Figure 3.18 help rationalize this observed structure? Explain. 100. The gasarrow_forward
- Help mearrow_forwardWrite Lewis structures for these ions. Show all valence electrons and all formal charges. (a) Amide ion, NH2-arrow_forwardDraw Lewis structures for the following compounds.(a) ammonia, NH3 (b) water, H2O(c) hydronium ion, H3O+ (d) propane, C3H8(e) dimethylamine, CH3NHCH3 (f) diethyl ether, CH3CH2OCH2CH3(g) 1-chloropropane, CH3CH2CH2Cl (h) propane-2-ol, CH3CH(OH)CH3(i) borane, BH3 (j) boron trifluoride, BF3Explain what is unusual about the bonding in the compounds in parts (i) and (j)arrow_forward
- Write Lewis structures for these ions. Show all valence electrons and all formal charges. (Q) Acetate ion, CH3COO-arrow_forwardWrite Lewis structures for the following molecules or ions. (Assign lone pairs, radical electrons, and atomic charges where appropriate.) (a) BrF3 (b) AsF5 (c) BI3 (d) AsF6−arrow_forwardAlthough I3- is a known ion, F3- is not. (a) Draw the Lewis structure for I3- (it is linear, not a triangle). (b) One of your classmates says that F3 - does not exist because F is too electronegative to make bonds with another atom. Give an example that proves your classmate is wrong. (c) Another classmate says F3- does not exist because it would violate the octet rule.Is this classmate possibly correct? (d) Yet another classmatesays F3- does not exist because F is too small to make bonds tomore than one atom. Is this classmate possibly correct?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





