
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
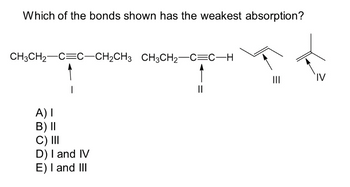
Transcribed Image Text:Which of the bonds shown has the weakest absorption?
CH3CH₂-C=C-CH₂CH3 CH3CH₂-C=C-H
1
A) I
B) II
C) III
D) I and IV
E) I and III
||
IV
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- S H-H 436 kJ/mol C-H 413 kJ/mol C=C 614 kJ/mol H-Cl 431 kJ/mol C-C 348 kJ/mol C=C 839 kJ/mol H-F 567 kJ/mol C-N 293 kJ/mol C=O 799 kJ/mol N-H 391 kJ/mol 358 kJ/mol 0=0 495 kJ/mol N-O 201 kJ/mol 485 kJ/mol C=O 1072 kJ/mol O-H 463 kJ/mol C-Cl 328 kJ/mol C=N 615 kJ/mol 0-0 146 kJ/mol C-S 259 kJ/mol N=N 418 kJ/mol F-F 155 kJ/mol Cl-CI 242 kJ/mol N=N 941 kJ/mol C=N 891 kJ/mol Estimate the enthalpy change (A Hrxn) of the following reaction for 2 moles of F2 using the bond energies above. HH H\ >H C=C H + F-F →→ kJ Question Help: Message instructor Submit Question JUL 29 H-C-C-H F F 648 231 W •arrow_forwardAssign 1,2 and 3 configuration S or Rarrow_forwardProvide the least stable conformation of the following compound. Cis/trans stereochemistry mattersarrow_forward
- Nonearrow_forwardH3C. H || IV Predict the correct splitting tree for circled hydrogen in the structure below. Br A) I B) II C) III D) IVarrow_forward= III Which of the following line drawings represents the correct stereochemistry of molecule shown in the Newman projection below? I H. H CH3 CH 3 H OCH3 OCH 3 OCH 3 II IV OCH 3 OCH3 III... A) I B) II C) III D) IVarrow_forward
- H3CO Predict the correct splitting tree for circled hydrogen in the structure below. H 11 IV H A) I B) II C) III D) IVarrow_forwardConsider this organic mechanism: 1 H + + H :0 : OH 2 X Click and drag to start drawing a structure.arrow_forwardChoose the correct representation of the structure using chair conformation. Br H ogs A C E H H H 5 H H oft H Br Br Br "I B D TI F H H H H H H Br Z Br Brarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY