
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
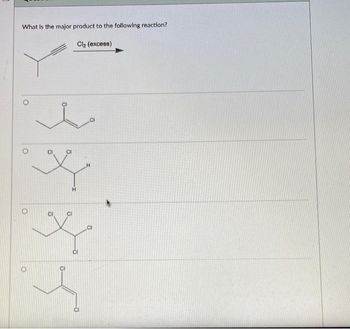
Transcribed Image Text:What is the major product to the following reaction?
Cl₂ (excess)
O
O
O
O
CI
CI
K
CI CI
K
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write a balanced equation to describe your observations. (Hint: This reaction is a double-displacement process.)arrow_forward2 HC= CH + 5 O2 > 4 co2 + 2 h2o What is the energy of the reaction and is it exo- or endothermic?arrow_forwardBalance the chemical equation below. Be(C2H3O2)2 + Al2(CO3)3 → BeCO3 + Al(C2H3O2)3 What type of reaction is this?arrow_forward
- Determine the 2 products yielded by the following reaction.arrow_forwardWhich of the compounds below is/are possible product(s) for the following reactions? Br CH;CH,OH %3D II IV O l and II O l and II O I, II, II, and IV O l and IVarrow_forwardK2CO3 + 2HCI -> H2O + 2KCI Determine the theoretical yield of CO2 if you start with 1000 g of K2CO3?arrow_forward
- Chloral hydrate is broken down in the body after several hours. One reaction is oxidation to trichloroethanoic acid. Complete the equation for this reaction below. Cl₂CCH(OH)2 + [0]arrow_forwardMatch each reaction or description to the change in entropy. CaCl₂ (s) + H₂O(l) CaO(s) + 2 HCl(g) Raindrops form in a cloud freezing water to form ice Frost forms on the windshield of your car Air is pumped into a tire CO₂(g) + H₂O(l) ----> H₂CO3(aq) | SOz(g) + CaO(s) -----> CaSO3(s) Ba2+ (aq) + 2F (aq) 2HgO (s) C₂H4(g) + H₂(g) ---> C₂H6 (g) boiling water to form steam dissolution of solid KCl in water PCL 3 (g) + Cl₂(g) PCL5(g) Sugar dissolves in coffee 2NO₂ (g) --> N₂O4 (g) BaF₂ (s) --> |2Hg (1) + O₂ (g) ---> --> ----> Gasoline vaporizes in the carburetor of an automobile engine Moisture condenses on the outside of a cold glass melting ice to form water mixing of two gases into one container seperating solute particles throughout a solvent Ni (s) + 2 HCl(aq) H₂(g) + NiCl₂(aq) a. As0arrow_forwardIdentify the oxidized atom in the following reaction: 2Fe2O3 (s)+3C (s)- 4Fe (s)+3CO2 (g) Fe O O OC None of the atomsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY