
Introduction to General, Organic and Biochemistry
11th Edition
ISBN: 9781285869759
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
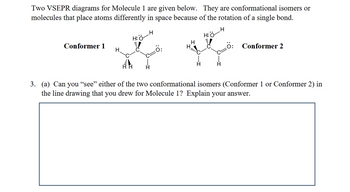
Transcribed Image Text:Two VSEPR diagrams for Molecule 1 are given below. They are conformational isomers or
molecules that place atoms differently in space because of the rotation of a single bond.
H
H
H:O
H:O
Conformer 1
H
H
H
Ö:
Conformer 2
3. (a) Can you “see” either of the two conformational isomers (Conformer 1 or Conformer 2) in
the line drawing that you drew for Molecule 1? Explain your answer.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- Following is a structural formula for cortisol (hydrocortisone). Draw a stereo-representation of this molecule showing the conformations of the five- and six-membered rings.arrow_forwardFollowing is a planar hexagon representation for one isomer of 1,2,4-trimethylcyclohexane. Draw the alternative chair conformations of this compound and state which of the two is more stable.arrow_forwardTwo VSEPR diagrams for Molecule 2 are given below. They are conformational isomers or molecules that place atoms differently in space because of the rotation of a single bond. Conformer 3 H H H H H H C4 Conformer 4 H 3. (b) Can you "see" either of the two conformational isomers (Conformer 3 or Conformer 4) in the line drawing that you drew for Molecule 2? Explain your answer.arrow_forward
- Perspective drawings of Chair and Boat Conformers fo Cyclohexane (C6H12). Note it is impossible to place all the carbons in the same plane without straining the bonds. Take two opposite carbons and pull both of them up to make one conformation adn then pull one of them down to make the other conformation. Can you inercovert one conformer int othe other without breaking any bonds? Explain why these represent conformers and not isomers.arrow_forwardClassify the pairs of molecules as not the same molecule, structural isomers, diastereomers, enantiomers or identical. Circle any molecules that are not achiral (a) (b) (c) Br... H CI H H Br H H3C Br H. CI Br H Br CI I I Br" "H H Br H CH3 Cr Brarrow_forwardIt is easy to imagine a cyclohexane as a flat hexagon and a lot of the time we draw it that way. Looking at 1,3,5-triethylcyclohexane we cannot tell the stability of the molecule from looking at the flat 2D drawing. Explain why we need to look at the 3D configuration and what conformation (axial,equatorial) would each of the three ethyl groups be in for the most stable configuration.arrow_forward
- There are four cis,trans isomers of 2-isopropyl-5-methylcyclohexanol:(a) Using a planar hexagon representation for the cyclohexane ring, draw structural formulas for the four cis,trans isomers.(b) Draw the more stable chair conformation for each of your answers in part (a).(c) Of the four cis,trans isomers, which is most stable? (Hint: If you answered thispart correctly, you picked the isomer found in nature and given the name menthol.)arrow_forwardFO 23: Identify and draw conformers. NH₂ NH2 NH2 NH2 NH2 NH2 1 NH2 NH2 4 2 3 (a) List any two structures that are related as conformers. Answer by listing exactly two numbers. (b) Draw one conformer of the new structure shown here: 1 and 3 (b) OH HO OHarrow_forwardPls help ASAP. Pls circle the final answerarrow_forward
- 4. Isomers and Stereocenters ( (a) Assign each set of molecules as constitutional isomers, conformational isomers, enantiomers, diastereomers, or identical. Please write the full name for the type. Molecules Type (b) H₂Ca HgC.. CH3 OCH₂ CH3 af OCH, SP H₂C**** H₂C H₂C NGOCH, H.C_OCKS OCH₂ CH₂ H₁C HC H*** CI H H CI Br H3C CH₂ Br (d) E) (0) For each of the following molecules, write the number of stereocenters and circle the center of any stereocenters. (i) (ii) H₂C (онт (eg 5) (b) -CH3 Isegna CH3 on mots eno H₂C CH3arrow_forwardFor 1,2-dichloroethane: (a) Draw Newman projections for all eclipsed conformations formed by rotation from 0 to 360° about the carbon-carbon single bond. (b) Which eclipsed conformation(s) has the lowest energy? Which has the highest energy? (c) Which, if any, of these eclipsed conformations are related by reflection?arrow_forwardWhich of the following is the highest energy chair conformer of the most stable isomer of 1-ethyl-2,4-dimethylcyclohexane? A | B II C) III IV = || ||| > INarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning