
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
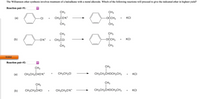
Transcribed Image Text:The Williamson ether synthesis involves treatment of a haloalkane with a metal alkoxide. Which of the following reactions will proceed to give the indicated ether in highest yield?
Reaction pair #1:
CH3
CH3
(a)
CI
CH;ĊOʻK*
-OČCH3
KCI
+
CH3
ČH3
CH3
CH3
OČCH3
KCI
CH3CI
CH3
(b)
-OK*
+
ČH3
Visited
Reaction pair #2:
CH3
CH3
CH;CH2CHOK*
CH3CH2CI
CH3CH2CHOCH2CH3
KCI
+
(a)
+
CH3
CH3
(b)
CH3CH2CHCI
CH;CH2OK*
CH3CH2CHOCH2CH3
KCI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the product (including stereochemistry) when the following substance reacts under thermal conditions. H H3C- H3C- H.arrow_forwardProvide the products of the following reactionsarrow_forward23) Which reaction will yield the following alkene as only elimination product? NaOEt A) Br E-3- Br NaOCH, B) KOIBU "Br KHSOJAT D) 24) What is the product of the following transformation? AgOH Br A) B) OH C) D) OH 25) Rank the following alkenes according to their heat of combustion from the LOWEST to the HIGHEST. II II IV A) II < IV < I< III B) III < I< IV < || C)I< || < IV < IIl D) II < IV < |I < Iarrow_forward
- Q3. 2-Bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant?arrow_forward+ H3C- CH₂ - CH2 - COR and have undergone McLafferty rearrangement and Ortho effect respectively. What are the possible fragments obtained as a result of these processes ? COOCH3 OH + (A) [R-CH₂-C (A) R-CH₂-CH₂ - CH₂ - COH @E OH]: [c C6H4 O and CO (B) H₂C=CROH and H₂C = CH₂ [2] : [ C₂H4O3 and CH4 (C) (D) H₂CCROH and C₂H4; [(C6H4O), CO and CH₂OH] RH, C₂H4 and CH₂CO; [(C₂H4O), CO and CH₂OH]arrow_forwardGive the major organic product(s) for the following reactionarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY