
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
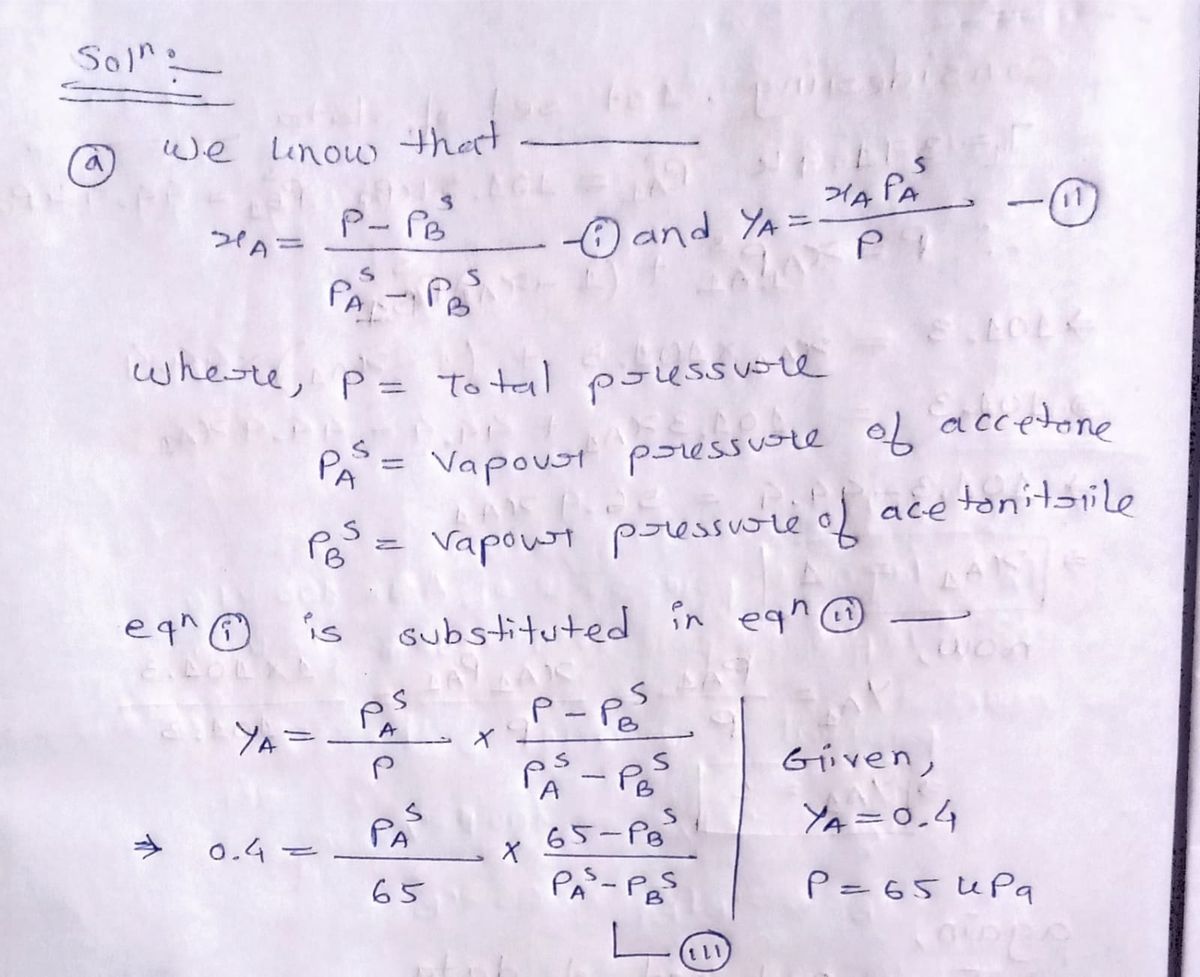
The vapour pressures of acetone (A) and acetonitrile (B) can be evaluated by the following Antoine equation.
where T is in K and P is in kPa. Assume that acetone and acetonitrile form ideal solutions. For a vapour phase consisting of 40% acetone and 60% acetonitrile, calculate the following:
(a) The dew point temperature and concentration of the first drop of liquid at 65 kPa (b) The dew point pressure and concentration of the first drop of liquid formed at 327 K
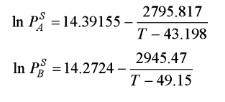
Transcribed Image Text:2795.817
T-43.198
2945.47
T - 49.15
In P = 14.39155-
In P = 14.2724-
Expert Solution
arrow_forward
Step 1
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Similar questions
- At approximately atmospheric pressure, methane gas is burned completely with 30% excess air. Both the air and methane enter the furnace at 30 °C saturated with water vapor, and the flue gases leave the furnace at 1500 °C. Calculate how much heat is lost from the furnace per mole of methane. In your solution, show analytically all calculations, including the analytical derivation of the number of moles of water vapor entering the furnace. Use steam tables to determine Psat, and compare your answer with the answer you would obtain using Antoine’s and Wagner’s equations. For the latter, you can determine A, B, C, D as in ** above.arrow_forwardIn an electrochemical cell, a metal anode lost 0.195 g while a total volume of 0.04592 L of hydrogen gas was produced. The temperature was 298 K and the barometric pressure was 768.1 mm Hg. At 298 K, the vapor pressure of water is 23.8 mm Hg. What is the molar mass of the metal?arrow_forwardAt a certain temperature and pressure, a 1.00 mol sample of argon gas is pumped into a 22.4 L rigid box that already contains 1.00 mol of nitrogen gas. We would expect the argon gas to: Group of answer choices decrease the total gas pressure in the box by a factor of 2 occupy the entire 22.4 L volume of the box. increase the total gas pressure in the box by a factor of less than 2. spread out into the box, but the actual volume occupied but he gas cannot be known without pressure information. occupy only 11.2 L of the box.arrow_forward
- Wet steam is water vapor containing droplets of liquid water. Steam quality defines the fraction of wet steam that is in the vapor phase. To dry steam (i.e., evaporate liquid droplets), wet steam (quality=0.89) is heated isothermally. The pressure of the wet steam is 4.8 bar and the flow rate of the dried steam is 0.488 m³/s. Determine the temperature (°C) at which the isothermal process occurs. Determine the specific enthalpy of the wet steam and the dry steam (kJ/kg). Determine the heat input (kW) required for the drying process.arrow_forward6) Calculate the absolute entropy for 1 mole of Ar gas (monatomic) with 22.4 liter volume at 273 K using Boltzmann's formula. What is the change in entropy if the temperature doubles while V is constant? What is the change in entropy if the volume doubles while T is constant? What is the change in entropy if both T and V double? (Note: To evaluate In(N!) use Stirling's asymptotic formula In(N!) ~ N In N – N ) S-k log W LVDWIG BOLTZMANN 18W-1906 DE FHILPAULA BOLTZMANN GER CARarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The