
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
(The pressure can be estimated from the Antoine equation and Raoult’s law to be164 mmHg.)
I need understanding how this pressure was calculated. Be thorough to the fullest.
![cal%20Processes,%204th%20Edition%20(%20PDFDrive%20).pdf
Copilot
Q168
The values of nout were determined from the known mole fractions of benzene and toluene in the outlet
streams and the calculated values of ny and nL. We do not know the feed-stream pressure and so we assume
that AH for the change from 1 atm to Pfeed is negligible, and since the process is not running at an unusually
low temperature or high pressure, we neglect the effects of pressure on enthalpy in the calculations of H₁
through H4. (The pressure can be estimated from the Antoine equation and Raoult's law to be 164 mm Hg.)
The heat capacity and latent heat data needed to calculate the outlet enthalpies are obtained from Tables B.1
and B.2.
-
Q Search
The formulas (including the APEx formulas) and values of the unknown specific enthalpies are given
below. Convince yourself that the formulas represent AH for the transitions from the reference states to the
process states.
H₁ =
=
(Cp)c,H₁1dT[=Enthalpy(“benzene”,10,50,“C”,“I”)] = 5.341 kJ/mol
50°C
H₂ = √ (C₂)CHT [=Enthalpy ("toluene", 10,50,"C","1")] = 6.341 kJ/mol
10°C
(Cp)C6H6(v)dT
50°C
H4 =
VITY EXERCISE
10°C
-80.1°C
H3 = √°C (Cp)C¡Há(1)ďT +(AĤv )C.H₂ (80.1°C) + +
10°C
451
110.62°C
of 695 Q
10°℃
80.1°C
[=Enthalpy("benzene", 10,80.1,"C","1") + Hv("benzene")
+Enthalpy("benzene",80.1,50,"C","g")] = 37.53 kJ/mol
The energy balance is
50°C
(C₂)CH(1)dT + (A‚)c‚µ‚(110.62°C) + fo
110.62°C
50°C
[=Enthalpy("toluene", 10,110.62,"C","1") + Hv("toluene")
+Enthalpy("toluene",110.62,50,"C","g")] = 42.94 kJ/mol
Q
Ση
Q = AH = Σn₁A₁ - Σnit₁ =
out
C
in
(Cp)C₂H₂(v) dT
Q=17.7 kJ
A gas emerges from a stack at 1200°C. Rather than being released directly to the atmosphere, it can
he nassed through one or several heat exchangers and the heat it loses can he nut to use in a variety
D
Ka
€
ENG](https://content.bartleby.com/qna-images/question/9ba88ecd-654b-4f6b-9783-e93c33ca6a16/fd12b65a-6603-4aa8-82fc-881b9d1b9602/sielfl4_thumbnail.jpeg)
Transcribed Image Text:cal%20Processes,%204th%20Edition%20(%20PDFDrive%20).pdf
Copilot
Q168
The values of nout were determined from the known mole fractions of benzene and toluene in the outlet
streams and the calculated values of ny and nL. We do not know the feed-stream pressure and so we assume
that AH for the change from 1 atm to Pfeed is negligible, and since the process is not running at an unusually
low temperature or high pressure, we neglect the effects of pressure on enthalpy in the calculations of H₁
through H4. (The pressure can be estimated from the Antoine equation and Raoult's law to be 164 mm Hg.)
The heat capacity and latent heat data needed to calculate the outlet enthalpies are obtained from Tables B.1
and B.2.
-
Q Search
The formulas (including the APEx formulas) and values of the unknown specific enthalpies are given
below. Convince yourself that the formulas represent AH for the transitions from the reference states to the
process states.
H₁ =
=
(Cp)c,H₁1dT[=Enthalpy(“benzene”,10,50,“C”,“I”)] = 5.341 kJ/mol
50°C
H₂ = √ (C₂)CHT [=Enthalpy ("toluene", 10,50,"C","1")] = 6.341 kJ/mol
10°C
(Cp)C6H6(v)dT
50°C
H4 =
VITY EXERCISE
10°C
-80.1°C
H3 = √°C (Cp)C¡Há(1)ďT +(AĤv )C.H₂ (80.1°C) + +
10°C
451
110.62°C
of 695 Q
10°℃
80.1°C
[=Enthalpy("benzene", 10,80.1,"C","1") + Hv("benzene")
+Enthalpy("benzene",80.1,50,"C","g")] = 37.53 kJ/mol
The energy balance is
50°C
(C₂)CH(1)dT + (A‚)c‚µ‚(110.62°C) + fo
110.62°C
50°C
[=Enthalpy("toluene", 10,110.62,"C","1") + Hv("toluene")
+Enthalpy("toluene",110.62,50,"C","g")] = 42.94 kJ/mol
Q
Ση
Q = AH = Σn₁A₁ - Σnit₁ =
out
C
in
(Cp)C₂H₂(v) dT
Q=17.7 kJ
A gas emerges from a stack at 1200°C. Rather than being released directly to the atmosphere, it can
he nassed through one or several heat exchangers and the heat it loses can he nut to use in a variety
D
Ka
€
ENG
Transcribed Image Text:*Elementary Principles of Chemic X +
ples%20of%20Chemical%20Processes,%204th%20Edition%20(%20PDFDrive%20).pdf
|
loud a
- 8.4-4
cyclopedia
Solution Basis: 1 mol Feed
rator
Ask Copilot
+
450 of 695
via capy changes for the unce stups.
college/felder
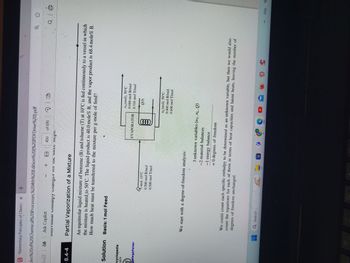
Partial Vaporization of a Mixture
An equimolar liquid mixture of benzene (B) and toluene (T) at 10°C is fed continuously to a vessel in which
the mixture is heated to 50°C. The liquid product is 40.0 mole% B, and the vapor product is 68.4 mole% B.
How much heat must be transferred to the mixture per g-mole of feed?
We start with a degree-of-freedom analysis:
mol, 10°C
0.500 mol B/mol
0.500 mol T/mol
Q Search
A
EVAPORATOR
QD
0000
ny(mol), 50°C
0.684 mol B/mol
0.316 mol T/mol
QU)
(mol), 50°C
0.400 mol B/mol
0.600 mol T/mol
3 unknown variables (ny, nL, Q)
-2 material balances
−1
-1 energy balance
= 0 degrees of freedom
We could count each specific enthalpy to be determined as an unknown variable, but then we would also
degrees of freedom unchanged.
count the equations for each of them in terms of heat capacities and latent heats, leaving the number of
Bal
Q 16
ENG
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 2 images

Knowledge Booster
Similar questions
- True or false: Osmotic pressure can be explained by the difference in fugacitiesarrow_forwardEstimate the Reynold's Number for 100% glycerol at 60°C flowing at a velocity of 0.3 m/s. The density of glycerol at 60 C is 1240 kg/m3. The diameter of the pipe is 15 mm. Is the flow turbulent or laminar?arrow_forward2.8. *The deepest point in the oceans of the world is believed to be in the Marianas Trench, southeast of Japan; there the depth is about 11 000 m. What is the pressure at that point?arrow_forward
- 2.5. Most swimmers find the pressure at a depth of about 10 ft painful to ears. What is the gauge pressure at this depth in psig and kPa?arrow_forwardIn assignment 3 I gave you the following expression for the pressure drop for turbulent flow through a horizontal smooth pipe of constant diameter at the special condition to which the Blasius equations applies: 0.25 L 0.75 1.75 AP = 0.241. BONUS Derive this equation. μ D4.75arrow_forward5arrow_forward
- 2. A clarifier-thickener system as show below is used to separate solids and liquids. Assume the density of solid is the same as water. Given the known quantities in the table below. Please fill in all missing numbers the table. A' E Stream A B Flow rate (Q, L/s) Solid (C, mg/L) 100 2500 90 10 6000 с D E A' 160arrow_forwardWhat pressure is exerted on the bottom of a 0.5m wide by 0.9 m long gas tank that can hold 50kg of gasoline by the weight of the gasoline in it when it is full?arrow_forward1. FA Newtonian fluid is flowing between two large parallel plates in the z direction. The top plate is moving in the positive z direction at a constant velocity Vo. The bottom plate is stationary. The velocity profile this flow is given by V(y) = V₁ (a) (b) (77) where 2H is the distance between the two plates. y is the vertical coordinate from the center plane, and is the velocity profile as a function of y. Draw a schematic of the flow system with the appropriate coordinate system. For plates with a total width W in the x-direction and length L in the z-direction, derive an expression for the volumetric flow rate Q through the plates.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The