
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
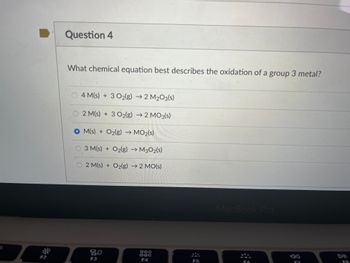
Transcribed Image Text:Question 4
What chemical equation best describes the oxidation of a group 3 metal?
4 M(s) + 3 O2(g) → 2 M₂O3(s)
2 M(s) + 3 O2(g) → 2 MO3(s)
OM(s) + O2(g) → MO₂(s)
01.0
3 M(s) + O2(g) → M302(s)
2 M(s) + O2(g) → 2 MO(s)
80
F3
000
000
F4
F5
MacBook Pro
FO
FO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Step by step please. also have to answer A-Darrow_forwardTin is smelted by heating tin (iv) oxide in the presence of carbon, releasing carbon monoxide as a by product. How many moles of carbon monoxide are produced from the reduction of 3.50 moles of tin (iv) oxide? Balance the reaction equation and show your work.arrow_forwardeduced. If it is not, select No and leave the following boxes blank. Express your answers as a chemical formulas. Omit st Sc(s) + 3Ag (ag) 3 Ag(s) + Sc+ (aq) a. Redox? Oxidizing Agent Reducing Agent Substance Oxidized Substance Reduced b. HI(g) + NH3 (9) → NH,I(s) Oxidizing Agent Redox? Reducing Agent Substance Oxidized Substance Reduced SiBr. (1) + 2H2 0(1) → 4HB1(aq) + SiO2 (s) с. Oxidizing Agent Redox? Reducing Agent Substance Oxidized Substance Reducedarrow_forward
- Decide whether each chemical reaction in the table below is an oxidation-reduction ("redox") reaction. If the reaction is a redox reaction, write down the formula of the reducing agent and the formula of the oxidizing agent. S₂ (s) + 4Pb (s) 4PbS₂ (s) 70₂(g) + 2CH₂CH₂(g) → 4CO₂(g) + 611₂0 (g) 3 HSO (aq) + OH¯(aq) → SO² (aq) + H₂0 (1) redox reaction? yes reducing agent: oxidizing 0 agent: redox reaction? yes reducing agent: oxidizing agent: 0 redox reaction? yes reducing agent: oxidizing □ agent: no no no X Śarrow_forwardPlease answer the below 2 questionsarrow_forwardWhy is this equation not a redox reaction? Fe2(SO4)3(aq) + 3H2O(l)arrow_forward
- Classify each chemical reaction: Reaction Mg(s) + ZnI₂ (aq) → Mgl, (aq) + Zn (s) 2Na(s) + 2HCl(aq) → 2NaCl(aq) + H₂ (g) FeCl₂ (aq) + Bas (aq) → BaCl₂ (aq) + FeS (s) Type choose one choose one choose one X Śarrow_forwardFor each chemical reaction listed in the table below, decide whether the highlighted atom is being oxidized or r highlighted atom is being... reaction neither oxidized nor oxidized reduced reduced olo Cu SO 4(aq)+Zn(s) → ZnSO 4(aq)+Cu(s) 4 HF (9)+ SiO,(s) → SiF 4(9)+2 H,O(9) C(s)+O2(9) → CO,(9) N2(9)+3 H2(9) → 2 NH3(9)arrow_forwardClassifying Reactions and Balancing Chemical Equations Part A: For each of the chemical reactions that are listed below, balance the chemical equation and then state the type of chemical reaction. 1. - Cu (s) + O₂(g) → CuO (s) Reaction type: 2.H₂O ()→→O₂ (g) + H₂ (B) Reaction type: 3. Fe (s) + H₂O () Fe₂O3 (s) + H₂(g) Reaction type: 5. 4.H₂S (aq) + AsCl3 (aq) →→As2S3 (s) + HCl(aq) Reaction type: CaCO3 (s) → CO₂(g) + Reaction type: 6. C4H10O (g) + O₂(g) →→CO₂(g) + H₂O (c) Reaction type: 7. S8 (s) + Fe (s)→ FeS (s) Reaction type: CaO (s) 8. H₂SO4 (aq) +Al (s) →→Al2(SO4)3 (aq) + H₂(g) Reaction type: 9. H3PO4 (aq) + NH,OH (aq) Reaction type: (NH4)3PO4 (aq) + stei HOH (c) 2017-arrow_forward
- Specify which of the following are oxidation-reduction reactions, and if it is, identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance being reduced. If it is not, select No and leave the following boxes blank. Express your answers as chemical formulas. Omit states-of-matter. a. CH4 (g) + 302 (g) → CO₂(g) + 2H₂O(g) Redox? Oxidizing Agent Reducing Agent Substance Oxidized Substance Reduced b. 3AgNO3(aq) + La(s) → La(NO3)3(aq) + 3Ag(s) Redox? Oxidizing Agent Reducing Agent Substance Oxidized Substance Reduced Submit Answer Try Another Version item attempt remaining Cengage Learning Cengage Technical Support Previous Next> Email Instructor Save aarrow_forwardA chemist goes into the lab and performs an experiment in which a mixture of 8.47 grams of Zn and 6.03 g of sulfur are reacted. Before starting the experiment they perform the following calculations to predict the maximum (theoretical) yield of zinc sulfide that could be produced. First, write a balanced equation: Zn + 2 S → ZnS2 Second, check to see which reagent is limiting: 1 mole Zn 1 mole ZnS, 1 mole Zn 129.4 g ZnS, 1 mole ZnS, 8.47 g Zn × = 16.8 g ZnS, %3D 65.4 g Zn 1 mole ZnS, 129.4 g ZnS, 1 mole ZnS, 1 mole S 6.03 g S × = 12.2 g ZnS, 32.0 g S 2 moles S Since 6.03 g S produces the least amount of ZnS2, the S is limiting! They obtain an actual yield of 8.25 grams of zinc sulfide. Calculate their percent yield to one decimal place.arrow_forwardConsider the following redox reaction: Fe(s) + 2 HCl(aq) → FeCl2(aq) + H2(g) Which of the following statements are true? (Check all correct answers.) Group of answer choices The oxidation state of each Cl atom in FeCl2 is -2 Chlorine (Cl) is being reduced Fe(s) is being oxidized The oxidation state of iron in FeCl2 is +2 Hydrogen is being reduced The oxidation state of hydrogen in H2(g) is +1arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY