
Biochemistry
6th Edition
ISBN: 9781305577206
Author: Reginald H. Garrett, Charles M. Grisham
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
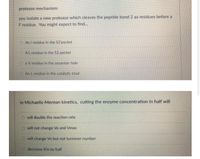
Transcribed Image Text:protease mechanism:
you isolate a new protease which cleaves the peptide bond 2 aa residues before a
F residue. You might expect to fınd...
O An I residue in the S2'pocket
AL residue in the S2 pocket
O aV residue in the oxyanion hole
An Lresidue in the catalytic triad
in Michaelis-Menton kinetics, cutting the enzyme concentration in half will
O will double the reaction rate
O will not change Vo and Vmax
will change Vo but not turnover number
decrease Km by half

Transcribed Image Text:chymotrypsin mechanism:
The rate limiting step of the reaction..
a. is the recognition of the substrate by the S1 pocket
b. happens before the system comes to steady state conditions
c. the release of the amino domain group of the peptide
d. is the formation of the acyl intermediate
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Using the ActiveModel for aldose reductase, describe the structure of the TIM barrel motif and the structure and location of the active site.arrow_forwardIn an enzymatic reaction: a. the enzyme leaves the reaction chemically unchanged. b. if the enzyme molecules approach maximal rate, and the substrate is continually increased, the rate of the reaction does not reach saturation. c. in the stomach, enzymes would have an optimal activity at a neutral pH. d. increasing temperature above the optimal value slows the reaction rate. e. the least important level of organization for an enzyme is its tertiary structure.arrow_forwardFigure 27.3 illustrates the response of R (ATP-regenerating) and U (ATP-utilizing) enzymes to energy charge. a. Would hexokinase be an R enzyme or a U enzyme? Would glutamine: PRPP amidotransferase, the second enzyme in purine biosynthesis, be an R enzyme or a U enzyme? b. If energy charge = 0.5: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low? c. If energy charge = 0.95: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low?arrow_forward
- Understanding the Mechanisms of Reactions Related to Transketolase The mechanistic chemistry of the acetolactate synthase and phosphoketolase reactions (shown here) is similar to that of the transketolase reaction (Figure 22.30). Write suitable mechanisms for these reactions.arrow_forwardAllosteric Regulation of Ribonucleotide Reductase by ATP and Deoxynucleotides Describe the underlying rationale for the regulatory effects exerted on ribonucleotide reductase by ATP, dATP, dTTP, and dGTP.arrow_forwardDistinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in animal muscle is a class 1 aldolase, which forms a Schiff base intermediate between substrate (for example. fructose-1, 6-bisphosphate or dihydroxyacetone phosphate) and a lysine at the active site (see Figure I8.12). The chemical evidence for this intermediate conies from studies with aldolase and the reducing agent sodium borohydride, NaBH4. Incubation of the enzyme with dihydroxyacetone phosphate and NaBH4 inactivates the enzyme. Interestingly, no inactivation is observed if NabH4 is added to the enzyme in the absence of substrate. Write a mechanism that explains these observations and provides evidence for the formation of a Schiff base intermediate in the aldolase reaction.arrow_forward
- Energetic of Fructose-1 ,6-bis P Hydrolysis (Integrates with Chapter 3.) The standard free energy change (G) for hydrolysis of fructose-1. 6-bisphosphate (FBP) to fructose-S-phosphate (F-6-P) and P: is -16.7 KJ/mol: FBP + H2O fructose-6-P + Pi The standard free energy change (G) for ATP hydrolysis is -30.5 KJ/mol: ATP + H2O ADP + Pj What is the standard free energy change for the phosphofructokinase reaction: ATP + fructose-6-P ADP + FBP b. What is the equilibrium constant for this reaction? c. Assuming the intracellular concentrations of [ATP] and (ADP] are maintained constant at 4 mM and 1.6 mM, respectively, in a rat liver cell, what will be the ratio of [FBP]/[fructose-6-P] when the phosphofructokinase reaction reaches equilibrium?arrow_forwardWhich of the following statements about inhibition is true? a. Allosteric inhibitors and allosteric activators are competitive for a given enzyme. b. If an inhibitor binds the active site, it is considered noncompetitive. c. If an inhibitor binds to a site other than the active site, this competitive inhibition. d. A noncompetitive inhibitor is believed to change the shape of the enzyme, making its active site inoperable. e. Competitive inhibition is usually not reversible.arrow_forwardAssessing the Effect of Active-Site Phosphorylation on Enzyme Activity (Integrates with Chapter 15.) The serine residue of isocitrate dehydroenase that is phosphorylated by protein kinase lies within the active site of the enzyme. This situation contrasts with most other examples of coa1ent modification by protein phosphorylation. where the phosphorylation occurs at a sate remote from the active site. What direct effect do you think such active-site phosphorylation might have on the catalytic activity of isocitrate dehydrogcn.ise? (Sec Barford, D., 1991. Molecular mechanisms for the control of enzymic activity by protein phosphorytation. Biochimica et Biophysica Acta 1133:55—62.)arrow_forward
- Why Do Anabolic and Catabolic Pathways Differ? Why is the pathway for the biosynthesis of a biomolecule at least partially different from the pathway for its catabolism? Why is the pathway for the biosynthesis of a biomolecule inherently more complex than the pathway for its degradation?arrow_forwardWhich of the following statements about the allosteric site is true? a. The allosteric site is a second active site on a substrate in a metabolic pathway. b. The allosteric site on an enzyme can allow the product of a metabolic pathway to inhibit that enzyme and stop the pathway. c. When the allosteric site of an enzyme is occupied, the reaction is irreversible and the enzyme cannot react again. d. An allosteric activator prevents binding at the active site. e. An enzyme that possesses allosteric sites does not possess an active site.arrow_forwardUnderstanding the Mechanism of the -Ketoglutarate Dehydrogenase Reaction tshe first step of the -Ketoglutarate dehydrogenase reaction involves decarboxylation of the substrate and leaves a covalent TPP intermediate. Write a reasonable mechanism for this reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Cengage Learning

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax