
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
thumb_up100%
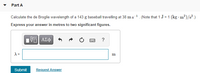
Transcribed Image Text:Part A
Calculate the de Broglie wavelength of a 143 g baseball travelling at 38 ms1. (Note that 1 J = 1 (kg · m²)/s².)
Express your answer in metres to two significant figures.
?
m
Submit
Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Ultraviolet radiation falls in the wavelength region of 1.00x10-8 to 1.00x107 meters. What is the frequency of ultraviolet radiation that has a wavelength of 1.00×10-8 m? Frequency = Submit Answer sec 1 Retry Entire Group 4 9 more group attempts remainingarrow_forwardUse the Heisenberg uncertainty principle to calculate Ax for a ball (mass 102 g, diameter 7.25 cm) with Av = 0.160 m/s. 1pts Submit Answer Incorrect. Tries 3/6 Previous Tries The uncertainty of the (above) ball's position is equal to what factor times the diameter of the ball? 1pts Submit Answer Tries 0/6arrow_forwardTry to solve it in 30 minutearrow_forward
- Calculate the de Broglie wavelength of a moving electron with kinetic energy equal to 13.759 electron volts (eV). Report your wavelength answer in angstrom units (Å). 1 eV = 1.60218×10-19 J, 1 Å=1×10-10 meters.______ Åarrow_forward▾ Part A Using the equation E = (hcRH) E₂ = 2 n Submit calculate the energy of an electron in the hydrogen atom when n = 2. Express your answer in joules to three significant figures. = (-2.18 x 10-18 J) ΤΟ ΑΣΦ -18 J) (-2) ΑΣΦΑΛΗ Request Answer Ú? Jarrow_forwardUse the References to access important values if needed for this question. A local AM radio station broadcasts at a frequency of 638 kHz. Calculate the wavelength at which it is broadcasting. Wavelength m (1 kHz = 10 sec ) %3D Submit Answerarrow_forward
- Please answer question 5 Part Aarrow_forwardPls help ASAP. Pls do both of the asked parts.arrow_forwardCalculate the wavelength of light emitted from a hydrogen atom when it undergoes a transition from the n=8 level to n=3. Enter the correct numeric value to 2.0% precision. Express scientific notation like 4.29E-15arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY