Normal No Spacing Heading Heading D. A- 三加 。、田 Paragraph Styles Part D. Energy Calculations The table below contains some information about four photons in the emission spectrum of Hydrógen. Perform the calculations needed to fill in the blanks in the table. You can refer to the diagram shown in Part D (Figure 5) if you find it helpful. Recall that for the Hydrogen atom, the following equation can be used 1 1 = R ,2 Where R = 1.097 x 107 m-1. %3D Table 1: Three photons in the Hydrogen emission spectrum. Photon A B C Initial n Final n 1 1 Energy (kJ/mol) 1230 Wavelength (nm) 123.3 Frequency (Hz) 7.32 x 1014 Spectral region Coleulete the ionization eneray (in k /moll and the related wavelenath for the
Normal No Spacing Heading Heading D. A- 三加 。、田 Paragraph Styles Part D. Energy Calculations The table below contains some information about four photons in the emission spectrum of Hydrógen. Perform the calculations needed to fill in the blanks in the table. You can refer to the diagram shown in Part D (Figure 5) if you find it helpful. Recall that for the Hydrogen atom, the following equation can be used 1 1 = R ,2 Where R = 1.097 x 107 m-1. %3D Table 1: Three photons in the Hydrogen emission spectrum. Photon A B C Initial n Final n 1 1 Energy (kJ/mol) 1230 Wavelength (nm) 123.3 Frequency (Hz) 7.32 x 1014 Spectral region Coleulete the ionization eneray (in k /moll and the related wavelenath for the
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter11: Atomic Theory :the Quantum Model Of The Atom
Section: Chapter Questions
Problem 103E
Related questions
Question
Would you be able to fill in table 1?

Transcribed Image Text:Mailings
Review
View
Help
ces
• A A
Aa v Ao
Normal
No Spacing Heading Heading
xA A-
田。
at
Paragraph
Styles
Part D. Energy Calculations
The table below contains some information about four photons in the emission spectrum
of Hydrógen.
Perform the calculations needed to fill in the blanks in the table. You can refer to the
diagram shown in Part D (Figure 5) if you find it helpful.
Recall that for the Hydrogen atom, the following equation can be used
1
R
%3D
Where R, = 1.097 x 107 m 1
Table 1: Three photons in the Hydrogen emission spectrum.
Photon
C
Initial n
6.
Final n
1
2
Energy (kJ/mol)
1230
Wavelength (nm)
123.3
Frequency (Hz)
7.32 x 1014
Spectral region
2. Calculate the ionization energy (in kJ/mol) and the related wavelength for the
hydrogen atom. (Hint: lonization energy is the energy required to remove an electron
completely from an atom.)
E Investigate
Expert Solution
Step 1 in
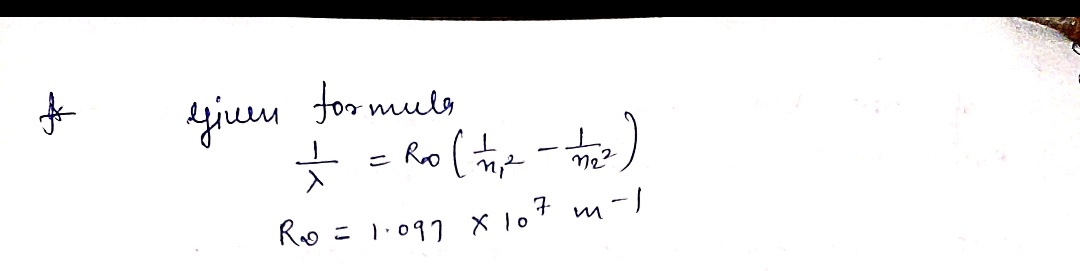
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning