
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
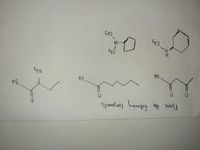
Transcribed Image Text:CH3
HQ.
HQ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A chemist prepares a solution of potassium iodide (KI) by measuring out 175. μmol of potassium iodide into a 200. mL volumetric flask and filling the flask to the mark with water. Calculate the concentration in mmol/L of the chemist's potassium iodide solution. Round your answer to 3 significant digits. mmol L 0 x10 X 3arrow_forwardthe Question 5 A graph that displays a straight line when the reciprocal of the concentration is plotted as a function of time. The graph has a slope of 0.176 and a y-intercept of 0.837. What is the concentration? (the answer should be entered with 3 significant figures; do not enter units; give answer in normal notation- examples include 1.23 and 12.3 and 120. and -123) Selected Answer O 5.68 Correct Anwer O 1.19 196arrow_forwardسلمہ مكمarrow_forward
- Nitrogen forms several gaseous oxides. One of them has a density of 1.25 g/L at 720 mm Hg and 152 deg C. What is the molecular formula for this compound? Hint Solve for molar mass first! Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a NO b NO2 N20 N202 16 NO3 e Fullscreen N30 8:48 PM a 中 4/11/2021arrow_forward4.05 kg + 567.95 g + 100.1 garrow_forwardBelow are 3 real life situations. Pick two (2) them and explain ALL the science involved in the scenario. This means describe things like what is happening to the gas/liquid/solid molecules or atoms, which principles or laws are at work, why what is happening is happening, and how you know. At sea level, a pot of water will boil at 100 °C. Suppose you wanted to boil the same amount of water high in the Rocky Mountains. What do you expect the temperature will need to be (generally speaking)? Why? A can of soda sits in a sunny spot on the window sill for several hours. It is then opened at the same time as a cold can of soda from the refrigerator. What happens and why? Weather balloons do not float out into space. They reach an altitude of about 15 miles above the surface (3x the height of Mt Everest) and they burst. Why?arrow_forward
- 7. The density of aluminum is 2.702 g/cm³. What is the final liquid level of water if 1.130 ounces of aluminum is dropped into a graduated cylinder containing 15.90 mL of water? (1 ounce = 28.3 g) A) 21.66 mL B) 17.08 mL C) 27.76 mL D) 47.95 mLarrow_forward3. A neon sign is constructed of a cylindrical glass tube with an inner diameter of 12.5 mm and a length of 4 ft. If the sign contains Ne at a pressure of 5.00 torr at 35°C, how many milligrams (mg) of Ne are in the sign? Vcylinder = mr21 25.4 mm = 1 in 12 in = 1 ft 1 ft³ = 28.32 Larrow_forwardminimum 6 sentences Future trends in green chemistry (arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY