
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
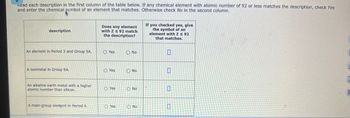
Transcribed Image Text:Kead each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes
and enter the chemical symbol of an element that matches. Otherwise check No in the second column.
description
Does any element
with Z ≤ 92 match
the description?
If you checked yes, give
the symbol of an
element with Z ≤ 92
that matches.
An element in Period 3 and Group 5A.
Yes
No
A nonmetal in Group 6A.
Yes
No
An alkaline earth metal with a higher
atomic number than silicon.
O Yes
O No
A main-group element in Period 4.
O Yes
No
O
☐
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the chemical symbol and the name of an element with three different isotopes with the corresponding mass (amu) and abundance (%): isotope 1 mass of 23.99 and abundance of 78.70, isotope 2 mass of 24.99 and abundance 10.13, isotope 3 mass of 25.98 and abundance of 11.17.arrow_forwardThe element zirconium has five naturally occurring isotopes. The masses (amu) and % abundances of the isotopes are given in the table below. Show your work to determine the average atomic mass. Isotope Mass % Abundance Zr-90 89.9043 51.40 Zr-91 90.9053 11.20 Zr-92 91.9046 17.10 Zr-94 93.9061 17.50 Zr-96 95.9082 2.80arrow_forward12, Which of the statements is not correct about the element germanium? Group of answer choices A, It is a metal. B, It is in group 4A. C,It has properties most similar to siicon. D, It is in period 4. 5. If two atoms have the same number of protons, but the different number of neutrons, they have the same: Group of answer choices A, chemical properties B, atomic mass C, mass number D, physical propertiesarrow_forward
- An element has the following natural abundances and isotopic masses: 78.99% abundance with 23.985 amu, 10.00% abundance with 24.986 amu, and 11.01% abundance with 25.983 amu. Calculate the weighted average atomic mass of this element. Use the correct number of significant figures in your answer. Identify the element, by name and symbol.arrow_forwardRead each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes and enter the chemical symbol of an element that matches. Otherwise check No in the second column. description An element in Period 3 and Group 3A. A semimetal in Group 4A. An element in the nitrogen family with a higher atomic number than silicon. A transition element in Period 6. Does any element with Z ≤ 92 match the description? OYes O Yes Yes O Yes O No O No O No O No If you checked yes, give the symbol of an element with Z ≤ 92 that matches. 0 U 7arrow_forwardIdentify these elements based on their locations in the periodic table. Give the symbol, not the name. period 6, group 13 (3A) period 5, group 12 (2B) period 2, group 18 (8A)arrow_forward
- Identify these elements based on their locations in the periodic table. Give the symbol, not the name.arrow_forwardThe atomic number indicates_ the total number of neutrons and protons in a nucleus the number of different isotopes of an element the number of neutrons in a nucleus O the number of atoms in 1 g of an element the number of protons or electrons in a neutral atomarrow_forwardRead each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes and enter the chemical symbol of an element that matches. Otherwise check No in the second column. description Does any element with Z ≤ 92 match the description? If you checked yes, give the symbol of an element with Z ≤ 92 that matches. An element in Period 4 and Group 1A. Yes No A semimetal in Group 8A. Yes No ☐ An element in the nitrogen family with a higher atomic number than boron. Yes No ☐ A main-group element in Period 4. Yes No ☐ ☑arrow_forward
- Read each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes and enter the chemical symbol of an element that matches. Otherwise check No in the second column. description An element in Period 2 and Group 5A. A nonmetal in Group 7A. An alkaline earth metal with a lower atomic number than rubidium. A transition element in Period 6. Does any element with Z≤ 92 match the description? Yes Yes Yes Yes No No No No If you checked yes, give the symbol of an element with Z ≤ 92 that matches. 0 X 0 0 5arrow_forwardQUESTION Read each description in the first column of the table below. If any chemical element with atomic number of 92 or less matches the description, check Yes and enter the chemical symbol of an element that matches. Otherwise check No in the second column. Does any element with zS 92 match the description? If you checked yes, give the symbol of an element with z S 92 that matches. description An element in Period 4 and Group 5A. O Yes O No Yes No A semimetal in Group 8A. An element in the nitrogen family with a higher atomic number than indium. Yes No Yes No A main-group element in Period 2. More Practice O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy Accessibili acer a sieon ed backspace delete %23 $ 3 4 5 6. 8 t earrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY