Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.SE: Something Extra
Problem 35AP: A 1, 2-cis disubstituted cyclohexane, such as cis-1, 2-dichlorocyclohexane, must have one group...
Related questions
Question
Illsutrate the Ring-flipping interconverts axial and equatorial hydrogens in
cyclohexane ?
Expert Solution
Step 1
Cyclohexane-
- Structure is-
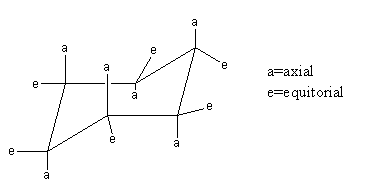
- In the conformational analysis cyclohexanes, cis and trans isomers are differential based on the position of substituents.
- The substituents may or may not be same. Only based on their relative positions in the ring they considered as cis and trans.

- These are positions in cyclohexane.
Step 2
Answer of given question-
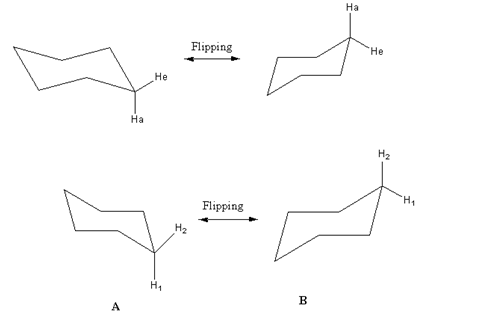
- In isomer A, H1 is the axial hydrogen and H2 is equatorial hydrogen.
- After flipping equatorial H2 in A goes to the axial position in B and also H1 in A goes to equatorial position in B.
- So equatorial hydrogens go to the axial position and axial hydrogens go to the equatorial positions after flipping of cyclohexane.
- Carbon-hydrogen bonds that are axial in first configuration become equatorial in other or vice versa. But the positions of their ‘up’ or ‘down’ does not change, remains as it is.
Step by step
Solved in 3 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning