
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
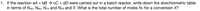
Transcribed Image Text:1. If the reaction aA + bB → cC + dD were carried out in a batch reactor, write down the stoichiometric table
in terms of NAo, NB0, Nco and Npo and X. What is the total number of moles N7 for a conversion X?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 1 images

Knowledge Booster
Similar questions
- For the following reaction set, define or propose two types of reactor systems to maximize product D selectivity, sketch the reactor system and describe clearly the input reactant concentrations and separation requirements if necessary. The unit of reaction rate is in units (mol/dm^3.sec) and concentration in units (mol/dm^3). (question on picture)arrow_forwardThe overall gas phase reaction 3A --> B + 2C occurs via the following sequence of elementary steps 2A <--> I1 + B k1,k-1 A + I1 --> 2I2 k2 I2 --> C k3 1. write an expression for the overall reaction rate, r, in terms of the rate step 3, r3 2. using the answer from 1, obtain an expression for the reaction rate, r, in terms of rate constants and concentrations of reactants and products, only use PSSH.arrow_forward1. Consider a gas-phase reaction, 2A - B +C + 3D with r = kC Initially 50% A and 50% inert are present. If the pressure and temperature remain constant, determine the final volume in terms of the initial and fractional conversion.arrow_forward
- Consider a gas-phase reaction, 2? → ? + ? + 3? with ?? = ??^2↓? Initially 50% A and 50% inert are present. If the pressure and temperature remain constant, determine the final volume in terms of the initial and fractional conversion.arrow_forwardGiven the following reactions, what is the enthalpy change for the reaction 3D -A+ 3B + 5C in kJ? (1) A- 3B + C, AH, = 100 kJ (2) A+C-D+B, AH, = -200 kJarrow_forwardConsider the reaction CaCO3(s)→CaO(s)+CO2(g).Estimate ΔG∘ and if they will be spontaneous for this reaction at 1475K, 1100K, & 295K. (Assume that ΔH∘ and ΔS∘ do not change too much within the given temperature range.)arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The