
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
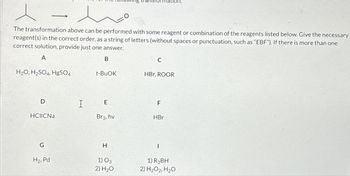
Transcribed Image Text:The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary
reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one
correct solution, provide just one answer.
A
B
H₂O, H₂SO4, HgSO4
D
-
HCECNa
G
H₂, Pd
I
t-BUOK
E
Br2, hv
H
1) 03
2) H₂O
C
HBr, ROOR
F
HBr
1
1) R₂BH
2) H₂O2, H₂O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Propose an efficient synthesis for the following transformation. 人人 The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as “EBF"). If there is more than one correct solution, provide just one answer. A B B HBr, ROOR D HBr G NaOH HC=CNa E CH3CH₂Br H conc. H₂SO4, heat с 1) R₂BH; 2) H₂O2, NaOH F H₂SO4, H₂O, HgSO4 1) LiAlH4; 2) H3O+arrow_forwardLast answer was wrong EDIarrow_forwardFropose an efficient synthesis for the following transformation: The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A B t-BUOK HBr, ROOR NazCr207, H2SO4, H2O HBr G H2, Lindlar's cat. 1) O3; 2) DMS TSCI, py dilute H2SO4 K 1) LIAIH4; 2) H3O* DMP or PCC conc. H,SO, heat 1) MeMgBr; 2) H3O*arrow_forward
- Identify the reagents you would use to perform the following transformation: The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagents in the correct order, as a string of letters (without spaces or punctuation, such as "EBF). If there is more than one correct solution, provide just one answer. A.H3O* B. Na,Cr207. H,SO,a. H20 C. NaČN D. 1) CO2 2) H3O* E. SOCI, F. NaCiarrow_forwardPropose an efficient synthesis for the following transformation: OH Ph Ph- -Ph Ph The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A 1) MeMgBr 2) H3O* 1) PhMgBr 2) H3O* 1) EtMgBr 2) H30* E F Na2Cr207, H2SO4. H2O H20, H2SO4, 1) R2BH 2) H2O2, NAOH HgSO4arrow_forward4. The following transformations require multiple steps. Show how to accomplish each transformation. You may use any necessary reagent containing 4 carbon atoms or fewer. Š Br NH₂ N + Brarrow_forward
- OH OH The transformation above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order for each transformation, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A t-BuOK D H2, Pt G B C HBr, ROOR conc. H2SO4 E F H2, Lindlar's cat. HBr H 1) BH3.THF; 2) H2O2, NaOH HC=CNa TsCl, pyridinearrow_forwardIdentify reagents that can be used to achieve the following transformation: H₂O, H₂SO4, HgSO4 The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A D 1) 03 2) DMS G سعد 1) xs NaNH2 2) H₂O B Br2 E H H₂, Lindlar's catalyst H t-BuOK с HC=CNa F 1) R₂BH 2) H₂O2, NaOH I H₂, Pdarrow_forwardCan you help me get the missing products. Drawn out in extended form please?arrow_forward
- OH HO The transformation above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order for each transformation, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A В C t-BUOK HBr, ROOR conc. H2SO4 D E F H2, Pt H2, Lindlar's cat. HBr G 1) BH3-THF; 2) H2O2, NaOH HC=CNa TSCI, pyridinearrow_forwardIdentify reagents that can be used to achieve the following transformation: HO, The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A H2O, H2SO4, H9SO4 Br2 HC=CNa F Mel H2, Lindlar's catalyst 1) R2BH 2) H2O2, NaOH 1) xs NaNH2 1) O3 H2, Pd 2) H20 2) DMSarrow_forwardIdentify reagents that can be used to achieve the following transformation: lo H. The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A В C H20, H2SO4, HgSO4 Br2 HC=CNa D E F 1) O3 1) R2BH 2) H2O2, NAOH H2, Lindlar's catalyst 2) DMS G H 1) xs NANH2 2) H20 t-BUOK H2, Pdarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY