
Concept explainers
Please refer to the images as much as you need to.
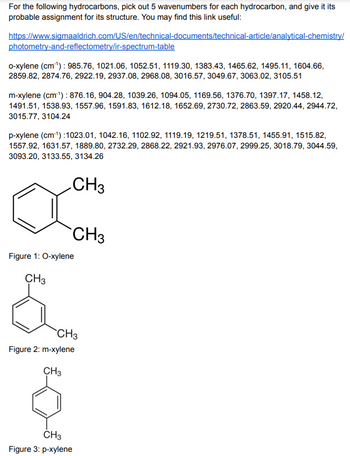
For the following hydrocarbons, pick out 5 wavenumbers for each hydrocarbon, and give it its probable assignment for its structure. You may find this link useful:
https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/analytical-chemistry/photometry-and-reflectometry/ir-spectrum-table
o-xylene (cm-1) : 985.76, 1021.06, 1052.51, 1119.30, 1383.43, 1465.62, 1495.11, 1604.66, 2859.82, 2874.76, 2922.19, 2937.08, 2968.08, 3016.57, 3049.67, 3063.02, 3105.51
m-xylene (cm-1) : 876.16, 904.28, 1039.26, 1094.05, 1169.56, 1376.70, 1397.17, 1458.12, 1491.51, 1538.93, 1557.96, 1591.83, 1612.18, 1652.69, 2730.72, 2863.59, 2920.44, 2944.72, 3015.77, 3104.24
p-xylene (cm-1) :1023.01, 1042.16, 1102.92, 1119.19, 1219.51, 1378.51, 1455.91, 1515.82, 1557.92, 1631.57, 1889.80, 2732.29, 2868.22, 2921.93, 2976.07, 2999.25, 3018.79, 3044.59, 3093.20, 3133.55, 3134.26
Step by stepSolved in 4 steps

- Please label the IR spectra.arrow_forwardThe volatile organic compounds (VOCs) produced by decomposing tissue and organs were studied in an effort to identify possib human-specific markers (PLoS ONE 2015 10(9): e0137341.). Of 452 VOCs isolated, six were found to be unique to humans, so th prove to be a useful tool for forensic chemists trying to determine the source of unidentified remains. There are two doublets in expected ¹H NMR spectrum of the following human-specific VOC. Identify the protons giving rise to each doublet. 1 2 H3C H3C- оооооооо 238 H 8 HH HH 3 HH CH3 5 7 Identify the two groups of protons that give rise to the doublets in the spectrum. CH3 8arrow_forwardPlease help me with this, I am trying to prepare for my final exam next week and I am just completely lostarrow_forward
- Determine the structure for the molecule that most likely produced these spectraarrow_forwardWhat alcohol compund is this based on this IR spectrum?arrow_forward5. Identify the molecule that corresponds to the IR spectra provided below. Indicate the significant band(s) with their approximate values that led to your conclusion. OH N HO. A B LOD TRANSMITTANCEI D 4000 3000 2000 HAVENUMBERI-L 1500 1000 500arrow_forward
- Please provide the C13NMR spectra for the following compounds:arrow_forwardPlease specify (and label) how many distinct signals would show up in the 1 HNMR spectra for each of the following substances. Give the integration ratio, splitting pattern, and roughly expected J-values.arrow_forwardinterpret/identify the spectroscopy peaks and its charecteristics (ex. C=O, 4H)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





