
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN: 9781259696527
Author: J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
Here are the enthapies for each compound. For acetone vapor, H(1)in = 35.7 kJ/mol and H(2)out= 32.0 kJ/mol. For nitrogen, H(3)out= -1.26 kJ/mol.
The written stuff on paper is additional information.
Transcribed Image Text:||!
PDF *Elementary Principles of Chemic X +
46°F
Sunny
File | E:/Elementary%20Principles%20of%20Chemical%20Processes,%204th%20Edition%20(%20PDF Drive%20).pdf
Draw
(T) Read aloud
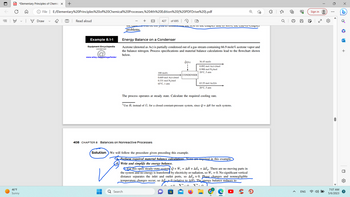
Example 8.1-1
Equipment Encyclopedia
condenser
www.wiley.com/college/felder
427 of 695
HA
Te Canice w UC for you to understand une rest of the chapter and to solve the cha-of-chapter
problems.
Energy Balance on a Condenser
Acetone (denoted as Ac) is partially condensed out of a gas stream containing 66.9 mole% acetone vapor and
the balance nitrogen. Process specifications and material balance calculations lead to the flowchart shown
below.
100 mol/s
0.669 mol Ac(v)/mol
0.331 mol N₂/mol
65°C, 1 atm
408 CHAPTER 8 Balances on Nonreactive Processes
Ô(J/s)
63.55 mol Ac(1)/s
20°C, 5 atm
The process operates at steady state. Calculate the required cooling rate.
Q Search
CONDENSER
2 Use Ĥ, instead of U; for a closed constant-pressure system, since Q=AH for such systems.
Solution We will follow the procedure given preceding this example.
36.45 mol/s
0.092 mol Ac(v)/mol
0.908 mol N₂/mol
20°C, 5 atm
■
Perform required material balance calculations. None are required in this example.
Write and simplify the energy balance.
For this open steady-state system + W, = AH + AÈk +AEp. There are no moving parts in
the system and no energy is transferred by electricity or radiation, so W, = 0. No significant vertical
distance separates the inlet and outlet ports, so AĖp ~0. Phase changes and nonnegligible
temperature changes occur, so AE 0 (relative to AH). The energy balance reduces to
r
P
<
Ơ
J
I
ENG
Sign in
100
7:07 AM
5/6/2023
•
la
+
O

Transcribed Image Text:Acetone (denoted as Ac) is partially condessed out of a gar
stream containing 66.9% acetone vapor and the balance nitrogen.
Calculate the required cooling rate.
[:liquid v-vapor & (5/5)
Joonol/s
6.669 mal Acco) not)
0.33/nal N₂ /no1
65°C 1 atm
Ас
Acid
& (J/s) 36.45 m³/s
B
Condenser
a
0.908 mol N₂t not
२००८ Sath
63.55 MASCA
20°G Safm
references. Ac (1, 20°C, 5 atr), N₂ lg 65° 4 lata)
2
bout Hout
66.9 H₁ 3.35 8/₂
63.550
33.1 33.1 M₂
O
/ Mol
Here is the process path for H
днь
Ac(1, 20°G 5atm) Stig Ac (1, 20°C, | atm) SHB Ach, 56°
Ac (0.65°C, latim)
Lath
Dita Ae (v. 56°, la tn)
M₁ = 0 Hpath
DH
A₁² Allis + Hibt OHICT OHLA
2
оинст дни
J
756°C
1₁² VAC(D() atn - 5 atm) + for ECRACIA THAHDAG
20°2
Show the calculations for Hi
also, show the paths for H₂ and Hy
and calculate the and My or well.
Also, use fabler Br and B₂
f
65
56
(Ep) dJ
Acl
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 4 steps

Knowledge Booster
Similar questions
- 2. Consider the P-x-y diagram at 300 K for A-B mixtures at right. (a) What are the vapor pressures of A and B at 300 K? (b) A 25 mol-% A vapor mixture is isother- mally compressed from 1.5 to 2.0 bar. Does a 2-phase mixture form, and if so, what is the mole fraction of A in each resulting phase? (c) Using data at the azeotrope, compute the van Laar parameters a and 3 for A-B mix- tures at this temperature. P (bar) 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 XA or YAarrow_forwardCalculate the fugacity of dry ice (CO₂ ice) at the triple point (T=216.55 °K, P-5.12673 bar). Assume that the thermal properties of the vapor phase of carbon dioxide are described by the Peng-Robinson equation of state. Express your result in bar and and round your numerical answer to one decimal digit.arrow_forward
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The