
College Physics
11th Edition
ISBN: 9781305952300
Author: Raymond A. Serway, Chris Vuille
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Topic Video
Question
Transcribed Image Text:6 of 6
<>
Constant
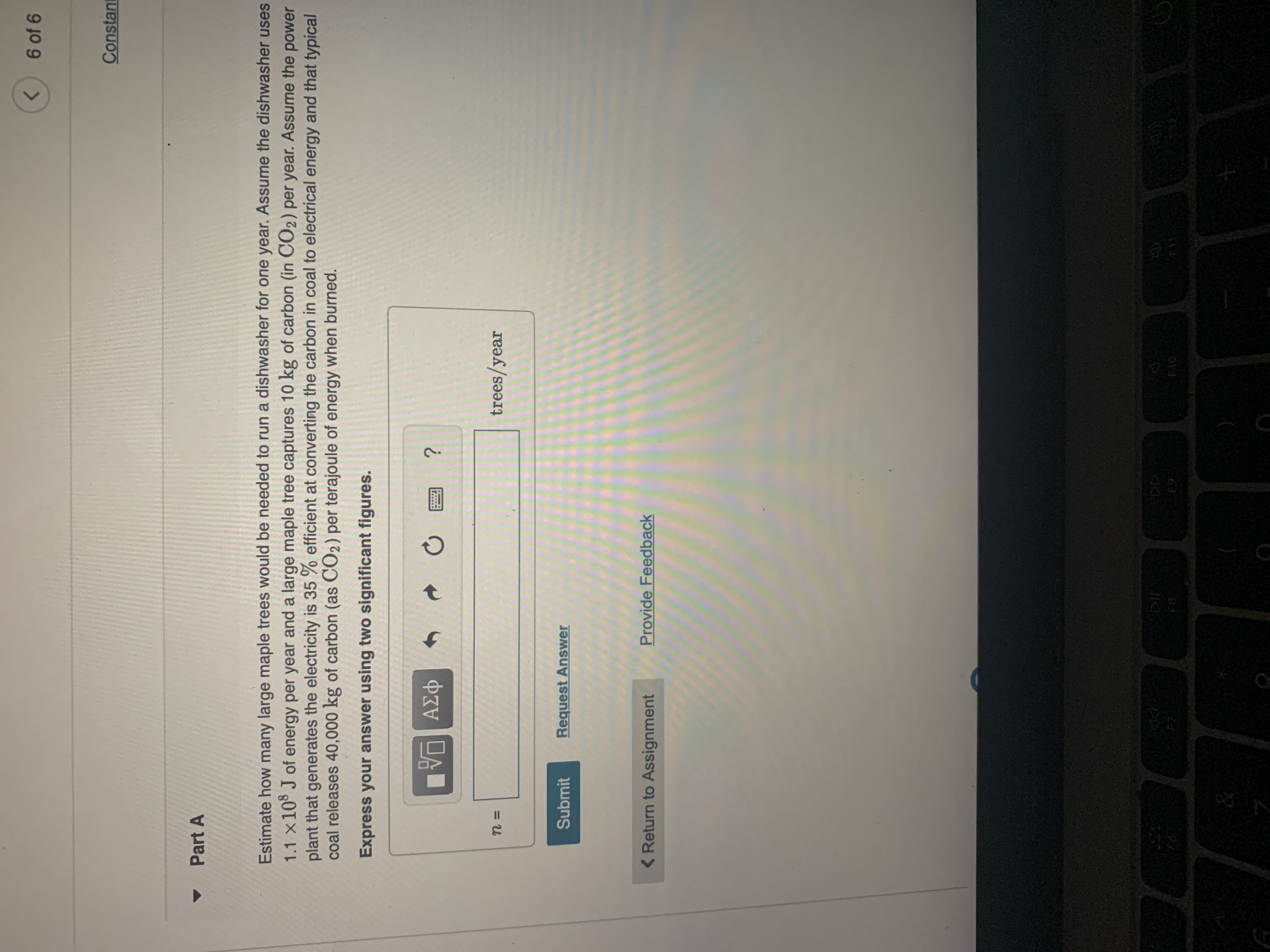
Part A
Estimate how many large maple trees would be needed to run a dishwasher for one year. Assume the dishwasher uses
1.1 x10 J of energy per year and a large maple tree captures 10 kg of carbon (in CO2) per year. Assume the power
plant that generates the electricity is 35 % efficient at converting the carbon in coal to electrical energy and that typical
coal releases 40,000 kg of carbon (as CO2) per terajoule of energy when burned.
Express your answer using two significant figures.
Φαν Α
trees/year
Submit
Request Answer
< Return to Assignment
Provide Feedback
F10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Consider the thermodynamic process, A->B->C->A shown above. The heat absorbed during A->B is 591J. If the change in internal energy during B->C is 4146J, What is the change in internal energy in SI units during C->A? Express only the number of your answer with 4 significant figures.arrow_forwardIn Arizona, photovoltaic (PV) panels can generate an average annual power of 1 MW/acre. If there are 311 sunny days per year in Arizona, but only 160 sunny days per year in Pennsylvania, how much average power (in MW/acre) could the same PV panels generate in Pennsylvania? Please include two decimal places in your answer. Note: assume that a cloudy day delivers 0 watts/acre of solar power. This is not true, but it simplifies the problem for us.arrow_forwardAn engine has a hot reservoir of 520K and a low-temperature reservoir of 330K. What is the theoretical efficiency of this engine? Answer in units of %arrow_forward
- Electric vehicles increase speed by using an electric motor that draws energy from a battery. When the vehicle slows, the motor runs as a generator, recharging the battery. Explain why this means that an electric vehicle can be more efficient than a gasoline-fueled vehicle.arrow_forwardA gas is compressed inside a cylinder. An average force of 80 N acts to move the piston 4.5 m. During the compression, 46 J of heat are conducted away from the gas. What is the change in internal energy of the gas? Calculate answer to one decimal.arrow_forwardA 2.0 m2 solar panel is fully illuminated by sunlight. Assume the sunlight has a power density of 0.10 J/cm2 -s. a) If the efficiency of the solar panel is 16%, how much electrical energy will it deliver in one hour? b) If this energy is stored in a battery, how long will it keep a 100 W incandescent lightbulb burning if the bulb has an efficiency of 2.6%. (Efficiency of the lightbulb is the light out - - 100 W - to the electrical power in.)arrow_forward
- A gas is compressed inside a cylinder. An average force of 10 N acts to move the piston 2.6 m. During the compression, 12 J of heat are conducted away from the gas. What is the change in internal energy of the gas? Calculate answer to one decimal.arrow_forwardbacon-cheddar-double-burger meal along with A 65.0-kg mountain climber just ate a Johnny Rocket's two peanut butter milkshakes, which have 5810 Calories (5810 kilocalories). Assume she can convert these calories into energy with 100% efficiency and use the fact that 1 kcal = 4186 J. Calculate how many times she could climb up Mt. Everest - the world's tallest mountain. The height of Mt. Everest is 8840 m.arrow_forwardA fuel-efficient car is able to convert the chemical potential energy of gasoline into translational kinetic energy at an efficiency of 78.4%. If there is 8742 kJ of chemical potential energy in one tank of gas, what is the amount of translational kinetic energy produced. How to manipulate formula eff=output/input x 100arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON