
Elements Of Electromagnetics
7th Edition
ISBN: 9780190698614
Author: Sadiku, Matthew N. O.
Publisher: Oxford University Press
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Determine the volume, in ft^3, of 2 lb of a two‐phase liquid–vapor mixture of Refrigerant 134A at 48°F with a quality of 50%. What is the pressure, in lbf/in^2?
Expert Solution
arrow_forward
Step 1
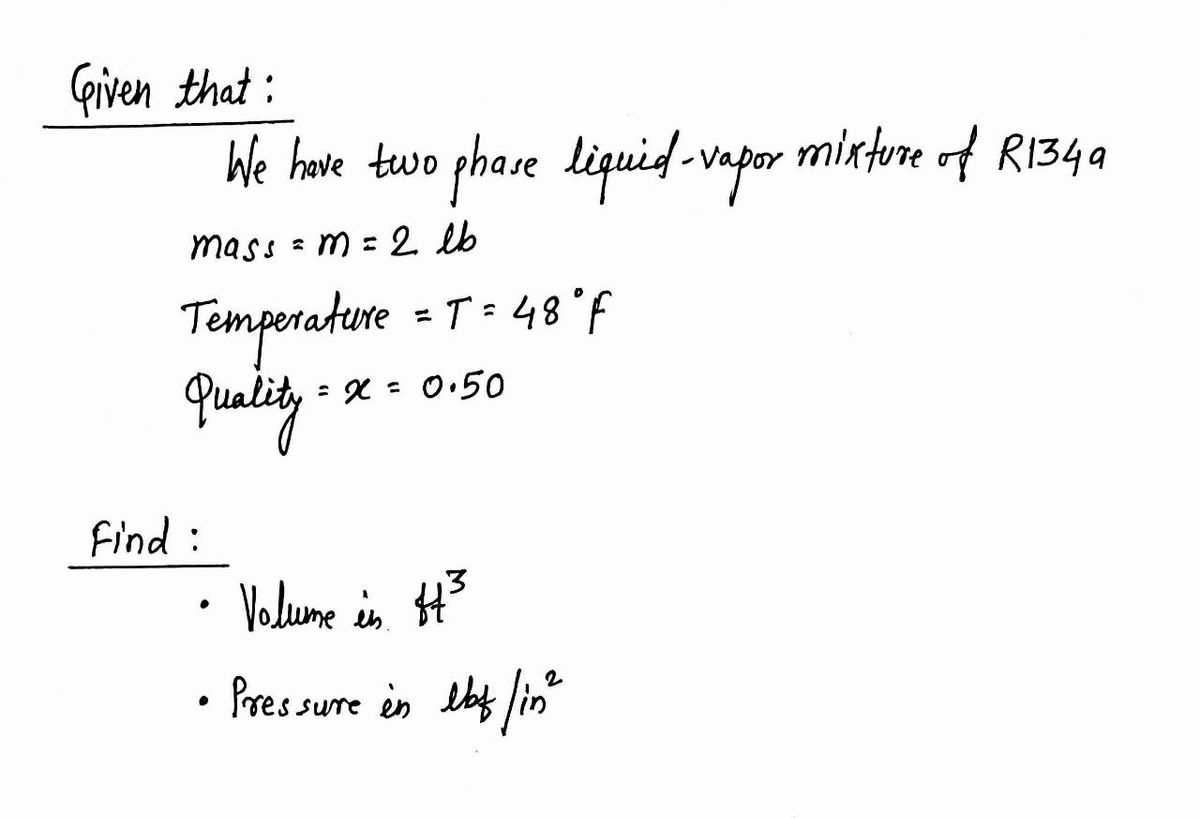
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- 2arrow_forwardA piston-cylinder assembly contains 2 lb of water, initially at 100 lbf/in.² and 400°F. The water undergoes two processes in series: a constant-pressure process followed by a constant volume process. At the end of the constant-volume process, the temperature is 300°F and the water is a two-phase liquid-vapor mixture with a quality of 60%. Neglect kinetic and potential energy effects. Determine the work and heat transfer for each process, all in Btu. Part A Determine the work for the constant-pressure process, in Btu. W12-38.05 Your answer is correct. Hint Part B Your answer is correct. Determine the heat transfer for the constant-pressure process, in Btu. Q12--300.366 Part C Hint W23- Btu Determine the work for the constant volume process, in Btu. Save for Later Btu Btu Attempts: 2 of 4 used Attempts: 1 of 4 used Attempts: 0 of 4 used Submit Answer Part D The parts of this question must be completed in order. This part will be available when you complete the part above.arrow_forwardA piston-cylinder assembly contains Refrigerant 22, initially a saturated vapor at 5 bar. The refrigerant undergoes a process for which the pressure-specific volume relationship is pv = constant to a final pressure of 20 bar. Kinetic and potential energy effects can be neglected. a. For your schematic, provide a rough sketch of your system, with arrows indicating direction of work and heat (i.e, in or out of the system) b. Determine the work and heat transfer for the process, each in (kJ/kg)arrow_forward
- A piston-cylinder assembly contains 2 lb of water, initially at 100 lbf/in.² and 400°F. The water undergoes two processes in series: a constant-pressure process followed by a constant volume process. At the end of the constant-volume process, the temperature is 300°F and the water is a two-phase liquid-vapor mixture with a quality of 60%. Neglect kinetic and potential energy effects. Determine the work and heat transfer for each process, all in Btu. Part A * Your answer is incorrect. Determine the work for the constant-pressure process, in Btu. W12= 38.65 Btuarrow_forwardFor H₂O, determine the specific volume at each of the indicated state, in m³/kg. (a) T = 600 ˚C, p = 20 MPa. (b) T = 80 °C, p = 20 MPa. (c) T = 60 °C, p = 2.5 MPa. Part A Determine the specific volume, in m³/kg, for state (a). m³/kg V = Part B Determine the specific volume, in m³/kg, for state (b). m³/kg V = Part C Determine the specific volume, in m³/kg, for state (c). m³/kg V =arrow_forwardAn oxygen gas R = 0.2598 KJ/kg°k and k = 1.395. If 4 kg of oxygen undergo a reversible non flow constant pressure process from initial volume =1.2 cubic meter and initial pressure = 690 kPa to a state where final temperature = 600°C. From the above statement: What is the initial temperature ? * 786.76°K 797.10 °K 876.50°K 796.77 °K Other: Determine the constant volume specific heat. * 0.6580 KJ/Kg-°K 0.6568 KJ/Kg-°K 0.5897 KJ/Kg- K 0.6577 KJ/Kg-°Karrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY