
Elements Of Electromagnetics
7th Edition
ISBN: 9780190698614
Author: Sadiku, Matthew N. O.
Publisher: Oxford University Press
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
thumb_up100%

Transcribed Image Text:3. The closed piston-cylinder assembly shown contains
Ammonia. The piston moves between two sets of stops. When
the piston is against the left stop, the enclosed volume is V₁
1.0 m³ and when the piston is against the right stop the
enclosed volume is V₁ = 2.5 m³. Atmospheric pressure is
1 bar. Heat is added so that the piston moves slowly to the
right. Sketch all processes on a p - v diagram and determine
the net work done during each process.
State 1: The piston is in contact with the left stop and P₁ = 0.8 bar
State 2: The piston first leaves the left stop
State 3: The piston first reaches the right stop
State 4: The pressure in the piston increases to P₂ = 5 bar
0
Ammonia
0
Expert Solution
arrow_forward
Step 1
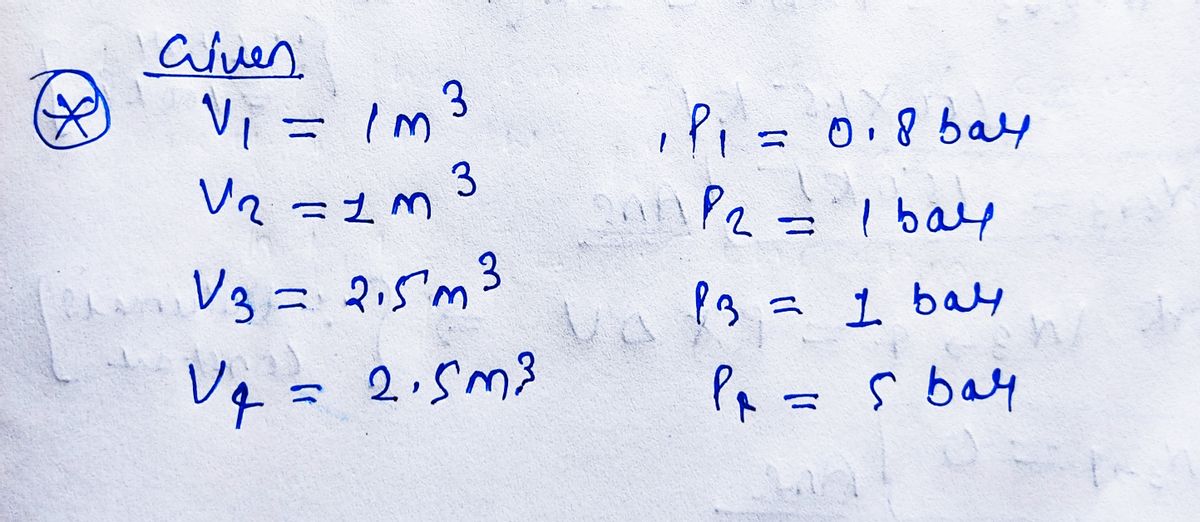
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- QUESTION 43 Carbon dioxide (molar mass 44 kg/kmol) expands reversibly in a perfectly thermally insulated cylinder from 3.7 bar, 220 UC to a volume of 0085 m If the initial volume occupied was 0.02 m calculate the adiabatic index to 1 decimal place. Assume nitrogen to be a perfect gas and take cv = 0 63 k J / k g K.arrow_forwardTask 4 A metal tank contains air at 20 C and 400 kPa. The tank volume is 5 m³. It is connected through a valve to another tank containing 7 kg of air at 40 C and 300 kPa. After 10 minutes, the valve is opened, and the whole system will reach thermal equilibrium with the surroundings (T= 25 C). Determine the volume of the second tank and the final pressure of air. Explain the relationships between system constants for a perfect gas.arrow_forwardA piston-cylinder contains gas initially at 3500 kPaa with a volume of 0.03 m². The gas is compressed during a process where pV¹25= C to a pressure of 8500 kPaa. The heat transfer from the gas is 2.5 kJ. Determine the change in internal energy, neglecting changes in kinetic and potential energies. [Ans. 141.9 kJ]arrow_forward
- Consider the series of processes shown in the p-V diagram below for a piston-cylinder system. Given the diagram information, determine the net work for the process from State 1 to State 3, in units of [kJ]. Specify whether the net work is into or out of the system, per our convention.arrow_forward* Your answer is incorrect. A piston-cylinder assembly contains 0.7 lb of propane. The propane expands from an initial state where p₁ = 60 lbf/in.² and T₁ = 70°F to a final state where p₂ = 10 lbf/in.² During the process, the pressure and specific volume are related by pv² = constant. Determine the energy transfer by work, in Btu. W = i 3.123 Btuarrow_forwardbe sure to follow sign convention.arrow_forward
- just part d answer options are 1.8kJ 1.3J 1.3kJ 1.8Jarrow_forwardOxygen (molar mass 32 kg/kmol ) expands reversibly in a cylinder behind a piston at a constant pressure of 3.102 bar. The volume initially is 0.0119 m and finally is 0.0327 m; the initial temperature is 18.24 °C. Calculate the heat supplied during the expansion in the correct unit to two decimal places. Assume oxygen to be a perfect gas and take the specific heat at constant pressure as = 0.917 kJ/kg Kand molar gas constant as = 8,314 J/ kmol Karrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY