
Introductory Chemistry: A Foundation
9th Edition
ISBN: 9781337399425
Author: Steven S. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Please correct answer and don't use hand raiting
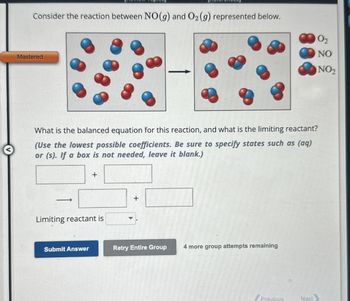
Transcribed Image Text:Consider the reaction between NO(g) and O2 (g) represented below.
Mastered
What is the balanced equation for this reaction, and what is the limiting reactant?
(Use the lowest possible coefficients. Be sure to specify states such as (aq)
or (s). If a box is not needed, leave it blank.)
+
Limiting reactant is
+
Submit Answer
Retry Entire Group 4 more group attempts remaining
Previous
Next
02
NO
NO2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- A 0.20 mol sample of magnesium burns in air to form 0.20 mol of solid MgO. What amount (moles) of oxygen (O2) is required for a complete reaction?arrow_forwardWhat is meant by a limiting reactant in a particular reaction? In what way is the reaction “limited”? What does it mean to say that one or more of the reactants are present in excess? What happens to a reaction when the limiting reactant is used up?arrow_forwardWrite the balanced chemical equation for the complete combustion of adipic acid, an organic acid containing 49.31% C, 6.90% H, and the remainder O, by mass.arrow_forward
- How many molecules of the sweetener saccharin can be prepared from 30 C atoms, 25 H atoms, 12 0 atoms, 8 S atoms, and 14 N atoms?arrow_forwardAmmonia can be formed from a combination reaction of its elements. A small fraction of an unreacted mixture of elements is illustrated in the following diagram, in which white spheres represent hydrogen atoms and blue spheres represent nitrogen atoms. The temperature is such that all species are gases. a Write and balance the equation for the reaction. b Which of the following correctly represents the product mixture? c Which species is the limiting reactant? Explain.arrow_forwardFor each of the following unbalanced reactions, suppose exactly 5.00 g of each reactant is taken. Determine which reactant is limiting, and also determine what mass of the excess reagent will remain after the limiting reactant is consumed. Na2B4O7(s) + H2SO4(o H3BOj(j) + Na2SO4(u CaC,(s) + H2O(/) Ca(OH)2(s) + C2H2(g) NaCl(s) + H2SO4(/> HCl(g) + Na2SO4(s) SiO2(s) + C(x) —> Si(/) + CO(g)arrow_forward
- High cost and limited availability of a reactant often dictate which reactant is limiting in a particular process. Identify the limiting reactant when the reactions below are run, and come up with a reason to support your decision. a. Burning charcoal on a grill: C(s)+O2(g)CO2(g) b. Burning a chunk of Mg in water: Mg(s)+2H2O(l)Mg(OH)2(aq)+H2(g) c. The Haber process of ammonia production: 3H2(g)+N2(g)2NH3(g)arrow_forwardTen mL of concentrated H3PO4 (91.7% by mass, d=1.69g/mL) was accidentally poured into a beaker containing 20.0 g of NaOH. Not all the NaOH was consumed. How many grams of NaOH were left unreacted? The equation for the reaction is H3PO4(aq)+3OH(aq)3H2O+PO43(aq)arrow_forwardConsider the hypothetical reaction between A2 and AB pictured below. What is the balanced equation? lf 2.50 moles of A2 are reacted with excess AB, what amount (moles) of product will form? If the mass of AB is 30.0 u and the mass of A2 are 40.0 u, what is the mass of the product? If 15.0 g of AB is reacted, what mass of A2 is required to react with all of the AB, and what mass of product is formed?arrow_forward
- In a common experiment in the general chemistry laboratory, magnesium metal is heated in air to produce MgO. MgO is a white solid, but in these experiments it often looks gray, due to small amounts of Mg3N2, a compound formed as some of the magnesium reacts with nitrogen. Write a balanced equation for each reaction.arrow_forwardThe pictures below show a molecular-scale view of a chemical reaction between H2 and CO to produce methanol, CH3OH The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. D Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forwardThe Hall process is an important method by which pure aluminum is prepared from its oxide (alumina, Al2O3 ) by indirect reaction with graphite (carbon). Balance the following equation, which is a simplified representation of this process. m:math>Al2O3(s)+C(s)Al(s)+CO2(g)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax