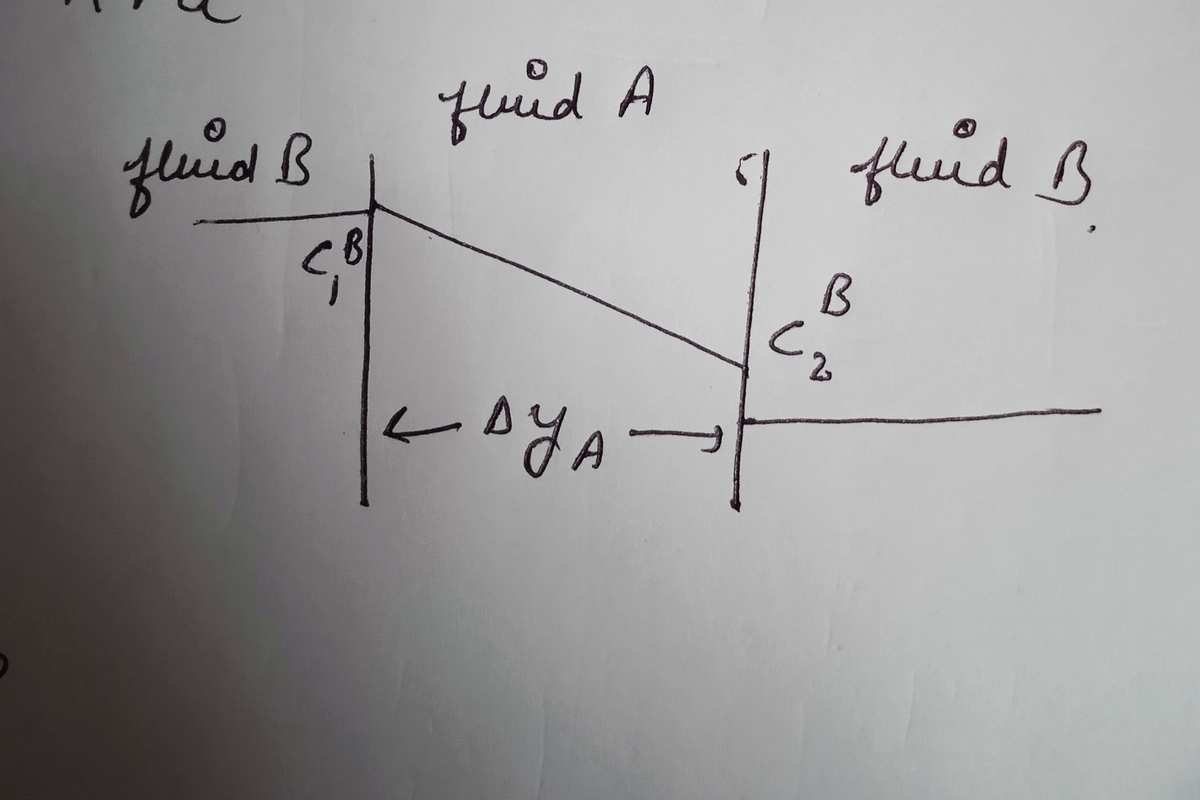
Consider a fluid layer A surrounded on both sides by a fluid, B. The fluid layer can be thought of as a membrane. A species S is diffusing across this membrane, and has concentrations c and c (in the fluid B) on the two sides of the membrane, as shown in Fig. 7.28. It often happens that the solubility of material S inside the membrane is different than its solubility in bulk solution B. We therefore define a partition coefficient k as concentration of S in material A (at equilibrium) (7.30) concentration of S in material B Hence k < 1 means that S is less soluble in the membrane, and k> 1 means S is more soluble. Write down an expression for the flux across the membrane in terms of c, c, DA (diffusion coefficient of S in A) and Aya. Sketch the concentration profile. What is the effective diffusion coefficient value with partitioning, De? Fluid A Fluid B Fluid B Concentration Concentration дуА Figure 7.28
Consider a fluid layer A surrounded on both sides by a fluid, B. The fluid layer can be thought of as a membrane. A species S is diffusing across this membrane, and has concentrations c and c (in the fluid B) on the two sides of the membrane, as shown in Fig. 7.28. It often happens that the solubility of material S inside the membrane is different than its solubility in bulk solution B. We therefore define a partition coefficient k as concentration of S in material A (at equilibrium) (7.30) concentration of S in material B Hence k < 1 means that S is less soluble in the membrane, and k> 1 means S is more soluble. Write down an expression for the flux across the membrane in terms of c, c, DA (diffusion coefficient of S in A) and Aya. Sketch the concentration profile. What is the effective diffusion coefficient value with partitioning, De? Fluid A Fluid B Fluid B Concentration Concentration дуА Figure 7.28
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter9: Heat Transfer With Phase Change
Section: Chapter Questions
Problem 9.26P
Related questions
Question

Transcribed Image Text:Consider a fluid layer A surrounded on both sides by a fluid, B. The fluid
layer can be thought of as a membrane. A species S is diffusing across this
membrane, and has concentrations c and c (in the fluid B) on the two sides
of the membrane, as shown in Fig. 7.28. It often happens that the solubility of
material S inside the membrane is different than its solubility in bulk solution
B. We therefore define a partition coefficient k as
concentration of S in material A (at equilibrium)
(7.30)
concentration of S in material B
Hence k < 1 means that S is less soluble in the membrane, and k> 1
means S is more soluble. Write down an expression for the flux across the
membrane in terms of c, c, DA (diffusion coefficient of S in A) and Aya.
Sketch the concentration profile. What is the effective diffusion coefficient
value with partitioning, De?

Transcribed Image Text:Fluid A
Fluid B
Fluid B
Concentration
Concentration
дуА
Figure 7.28
Expert Solution
Step 1
From Fick's first law of diffusion which states that diffusion flux of concentration goes from higher concentration to the lower concentration which can be written as
J= - D dc/dy
Here J is the diffusion flux
D is the diffusion coefficient or diffusivity and dc/dy is concentration gradient .
Step 2
Draw the diagram of the conentration profile for k<1 as,
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning