
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Please write answer in print and neat so I can read it please and thank you
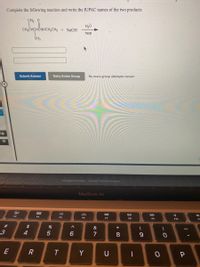
Transcribed Image Text:Complete the following reaction and write the IUPAC names of the two products.
CH3 0
H20
CH3CHCHÔNHCH2CH3 + NAOH
heat
CH3
Submit Answer
Retry Entire Group
No more group attempts remain
Cengage Learning | Cengage Technical Support
MacBook Air
80
888
DD
F3
F4
F5
F6
F7
FB
F9
F10
23
$
&
*
3
4
6.
7
E
R
Y
U
* 00
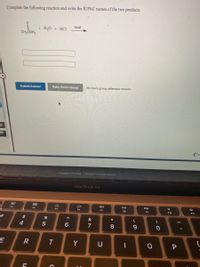
Transcribed Image Text:Complete the following reaction and write the IUPAC names of the two products.
heat
+ H2O + HCI
CH,ČNH,
Submit Answer
Retry Entire Group
No more group attempts remain
(Pre
Cengage Learning | Cengage Technical Support
MacBook Air
80
888
DII
DD
F3
F4
F5
F6
F7
F8
F9
F10
F11
23
2$
&
*
4
7
8.
R
T
Y
U
{
* 00
LL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Mla pnc.com/women Mail - Whi x O Grades for x Course Ho X G oxide cont X G Exercise 4.x E 4 - Molecu x O Bananas a X 1.84 x 10- x + envellum.ecollege.com/course.html?courseld 16985674&OpenVellumHMAC=a0b841caaee7feddab1623a6c83a92c0#10001 Review | Constants I Periodic Table Percent composition refers to the mass percent of each element in a compound: mass of element Part A mass percent = x 100% mass of compound > For example, the percent composition of water, H2O. is 11.2% hydrogen and 88.8% oxygen. Therefore, a 100-g sample of water contains 11.2 g of hydrogen atoms and 88.8 g of oxygen atoms. A hydrocarbon is a compound that contains mostly carbon and hydrogen. Calculate the percent composition (by mass) of the following hydrocarbon: CH22 Enter the percentages of carbon and hydrogen numerically to four significant figures, separated by commas. The periodic table will be useful when doing this problem. You can access a periodic table by clicking the "Tools" link in the upper right…arrow_forwardPlease give information about these chemicals.arrow_forwardI am just not understand this problem at allarrow_forward
- Explain if it is possible to combine different types of mass analyzer.arrow_forwardGive typed full explanation not a single word hand written otherwise leave itarrow_forwarda W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn 30.9 183.9 186.2 190.2 190.2 195.1 197.0 200.5 204.4 207.2 209.0 (210) (210) (222) 05 106 107 108 109 110 111 112 114 116 118 Ob Sg Bh Hs Mt Ds Uuu Uub 260) (263) (262) (265) (266) (271) (272) (277) (296) (298) (7) 60 61 62 63 64 65 66 67 68 69 70 71 Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 44.2 (147) 150.4 152.0 157.3 158.9 162.5 164.9 92 93 94 95 96 97 98 99 100 101 167.3 168.9 173.0 175.0 102 103 U Np Pu Am Cm Bk Cf F Es Fm Md No Lr 238) (237) (242) (243) (247) (247) (249) (254) (253) (256) (254) (257) sogna pee ou cacau pe busca o cus £(H၁)၁၀- шогеш аз моц моцѕ 01 смоле рәліп əsənу sәне иo pǝseq pod еш әсхә пол t be a part by a padd a test so be aarrow_forward
- There are 2 boxes for each reactant and product. Type the coefficient in the first box and the chemical formula in the second. Enter the chemical formulas in the order they appear in the problem. Each box must have a coefficient or formula. Type in a "1" if you did not change the coefficient. Type the formula in this format: CO_2. Omit the subscript "1" in the chemical formulas. This is case-sensitive. Carbon monoxide reacts with iron(III) oxide to produce iron and carbon dioxide.arrow_forwardPlease don't provide handwritten solution ....arrow_forwardPlease answer all question or leave for someone else to answer.arrow_forward
- 100% 41 Safari File Edit View History Bookmarks Window Help Wed 4:51 PM A uk.instructure.com My Questions | bartleby Course Details University of Kentucky - CHE 111 Lab - Fall19 - FRENCH: Quiz.. Calculate the Molarity of Sodium Hydroxide? | Yahoo Answers = CHE111-017-032 > CHE111 Sections 017 to 32 (Tuesdays): General Chemistry I Laboratory (Fall 2019) Fall 2019 Home Account Announcements Dashboard Question #: 11 Syllabus Course Details An aqueous solution containing 0.3845 g of KHP (KHC;H4O4) was titrated with a solution of sodium hydroxide, producing the following titration curve. What is the concentration of the NaOH solution? Courses Modules Titration of KHP with NaOH Piazza Groups 14 Grades 12 Calendar 10 Inbox 6. Help Volume NaOH (ml) DEC 18arrow_forwardYou have a 11.5 mg sample of blood that contains various proteins. Hemoglobin is the only protein in the sample containing Fe. Hemoglobin contains 3.83% by mass of a compound called heme (C₃₄H₃₂FeN₄O₄, MW = 616.49 g/mol). You find that your 11.5 mg blood sample contains 34.9 micrograms of Fe. What is the mass percent of hemoglobin in your protein sample? -don't prematureley roundarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY